

Background: Deucravacitinib, a first-in-class, oral, selective, allosteric tyrosine kinase 2 inhibitor, is approved in multiple countries for the treatment of moderate to severe plaque psoriasis. Deucravacitinib demonstrated efficacy in the phase 2 PAISLEY trial in patients with systemic lupus erythematosus (SLE) across multiple outcome measures, including a significantly higher rate of achieving ≥ 50% reduction from baseline in the Cutaneous Lupus Erythematosus Disease Area and Severity Index activity (CLASI-A) score (CLASI-50) at week 48 vs placebo [1]. Subgroup analyses from PAISLEY also showed higher rates of CLASI-50 response with deucravacitinib vs placebo in patients with SLE with acute cutaneous lupus erythematosus, subacute cutaneous lupus erythematosus (SCLE), or discoid lupus erythematosus (DLE) lesions [2]. Here, we report results from the phase 2 PAISLEY CLE trial of deucravacitinib vs placebo in patients with DLE and/or SCLE with or without SLE (NCT04857034).
Objectives: To assess efficacy and safety in patients with active DLE and/or SCLE with or without SLE treated with deucravacitinib 3 mg or 6 mg twice daily (BID) vs placebo.
Methods: Adults with a histologically confirmed clinical diagnosis of DLE and/or SCLE with active, moderate to severe cutaneous disease (CLASI-A score ≥ 8) were enrolled in this global, randomized, double-blind, placebo-controlled, phase 2 trial. Patients with SLE, according to the Systemic Lupus International Collaborating Clinics (SLICC) classification criteria, were limited to ≤ 50% of the population. Patients were randomized 1:1:1 to receive placebo or deucravacitinib (3 mg or 6 mg BID) for 16 weeks. At week 16, patients receiving placebo were rerandomized to deucravacitinib 3 mg or 6 mg BID until week 52; patients originally randomized to deucravacitinib continued treatment until week 52. The primary endpoint was mean percent change from baseline (CFB) in CLASI-A score at week 16. CLASI-50 and mean CFB in CLASI-A score at week 16 were among the secondary endpoints evaluated; exploratory endpoints included CFB in the patient-reported outcome (PRO) of skin pain at week 16, as measured by the skin pain visual analog scale (VAS), and mean percent CFB in CLASI-A score by visit to week 52. Achievement of ≥ 70% reduction from baseline in CLASI-A score (CLASI-70) at week 16 was assessed as a post-hoc analysis. Hypotheses were tested at a 2-sided 10% alpha level, with a P value of < 0.1 representing statistical significance. No adjustments for multiplicity were made in this phase 2 study. Exploratory endpoints were analyzed descriptively. Safety was also assessed.
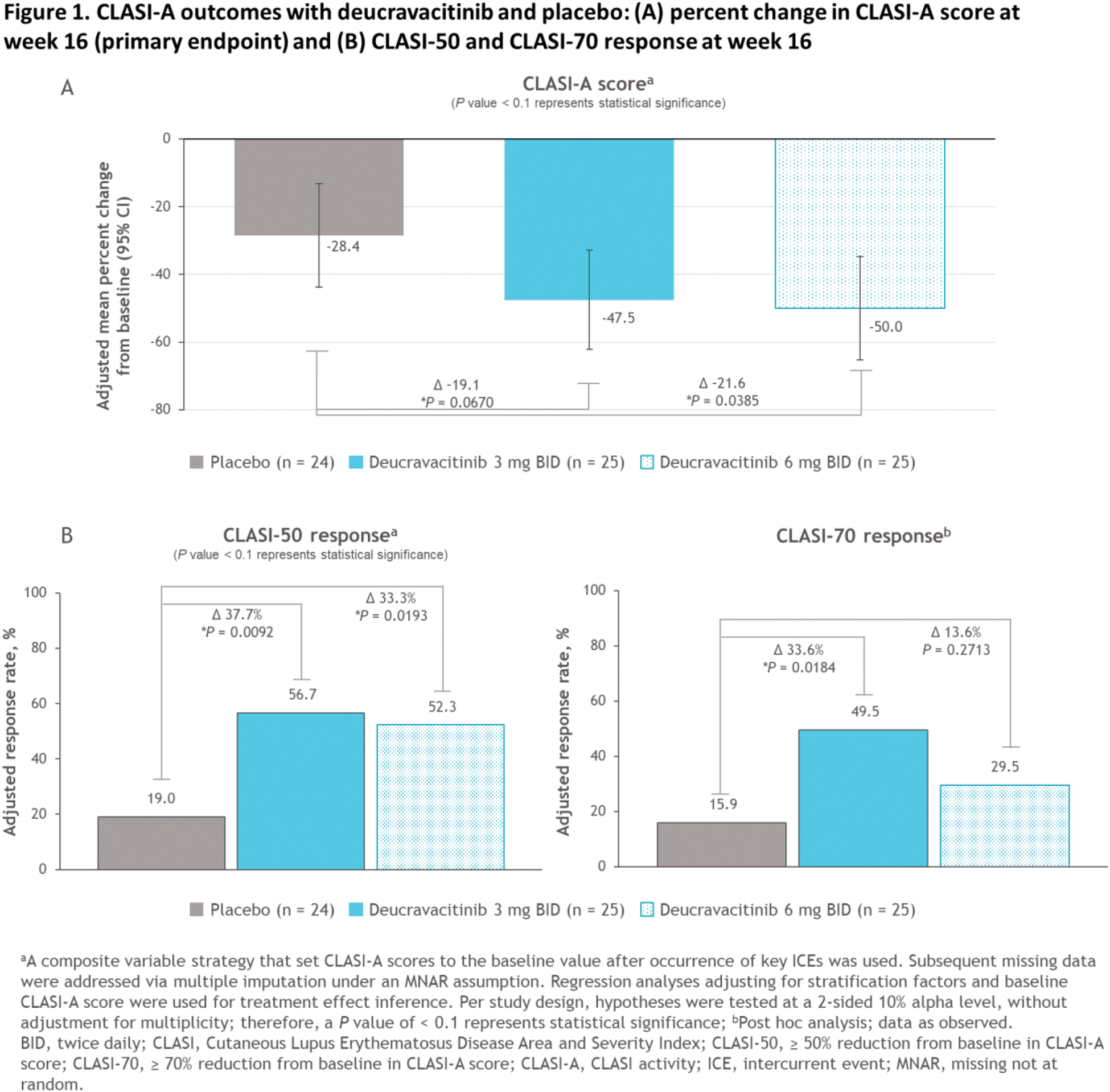
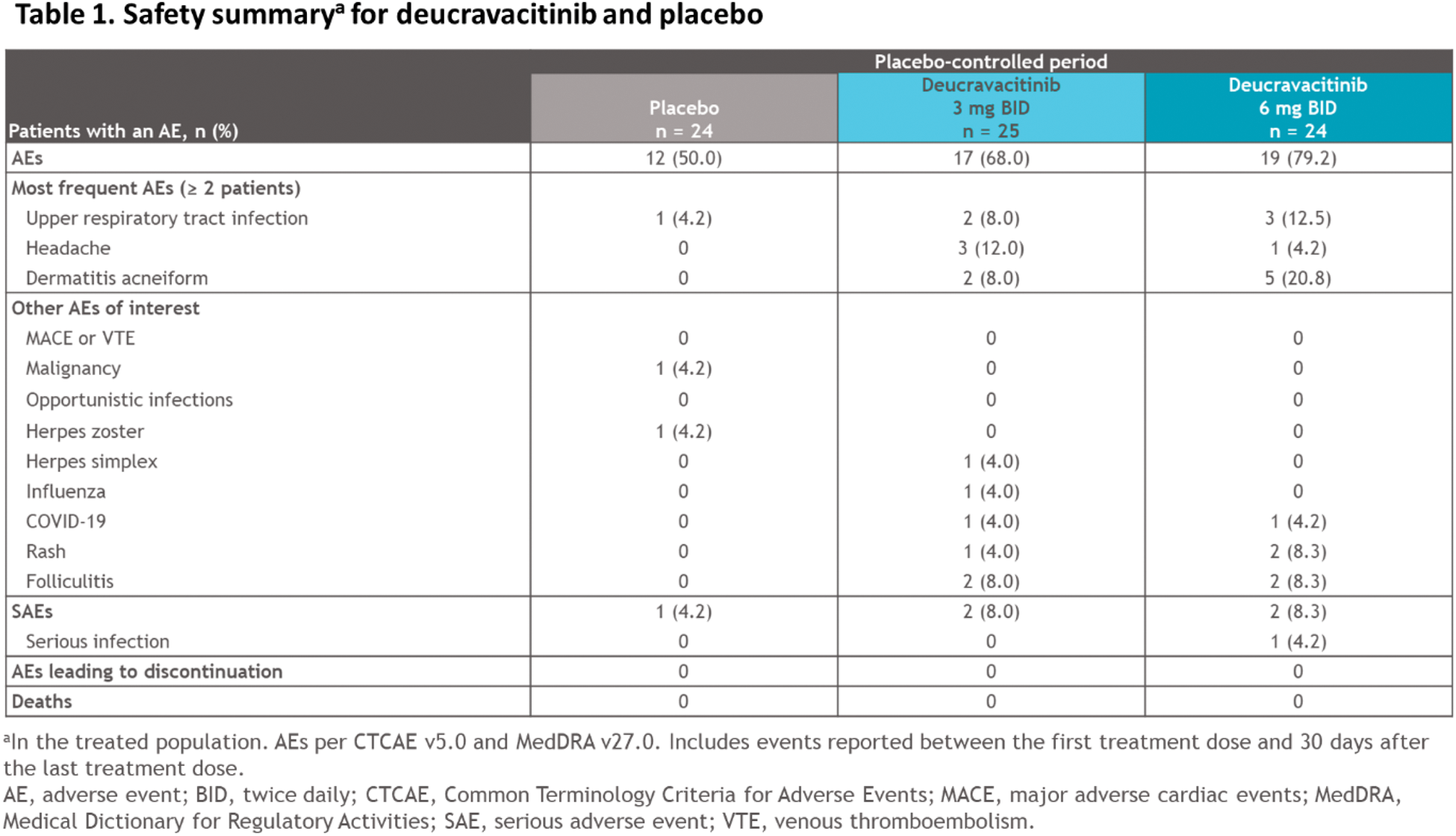
Results: Patients (N = 74) were randomized to placebo (n = 24), deucravacitinib 3 mg BID (n = 25), or deucravacitinib 6 mg BID (n = 25). Baseline demographics and disease characteristics were generally balanced across groups; most patients had moderate to severe skin disease. At screening, 41.7%, 32.0%, and 52.0% of patients in the placebo, deucravacitinib 3-mg BID, and deucravacitinib 6-mg BID groups, respectively, had SLE. Of randomized patients, 63 (85.1%) completed 16 weeks of treatment; no patients discontinued due to adverse events (AEs) or lack of efficacy. Deucravacitinib significantly improved mean percent CFB in CLASI-A score vs placebo (placebo, −28.4% vs deucravacitinib 3 mg BID, −47.5%, P = 0.0670, and 6 mg BID, −50.0%, P = 0.0385; Figure 1A), meeting the primary endpoint. Mean CFB in CLASI-A score was also significantly improved with deucravacitinib vs placebo (−5.3 vs −9.3, P = 0.0425, and −8.7, P = 0.0805). More patients treated with deucravacitinib vs placebo achieved CLASI-50 (19.0% vs 56.7% [ P = 0.0092] and 52.3% [ P = 0.0193]) and CLASI-70 (15.9% vs 49.5% [ P = 0.0184] and 29.5% [ P = 0.2713]; Figure 1B). Analysis of mean percent CFB in CLASI-A score over time showed numerical improvements with both deucravacitinib doses as early as 4 weeks, with a trend toward continued improvement to week 52. Skin pain was numerically improved with deucravacitinib vs placebo at week 16. No new safety signals were observed (Table 1). The most common AEs with deucravacitinib 3 mg and 6 mg BID at week 16 included upper respiratory tract infection, headache, and dermatitis acneiform (Table 1); most AEs were mild to moderate. No AEs led to treatment discontinuation in any arm. No cases of herpes zoster, major adverse cardiac events, venous thromboembolism, malignancy, or opportunistic infections occurred with deucravacitinib. No trends for clinically meaningful laboratory abnormalities were observed in any arm.
Conclusion: In patients with DLE and/or SCLE with or without SLE, statistically significant and clinically meaningful improvements were observed for the primary endpoint and different CLASI-A endpoints with deucravacitinib 3 mg and 6 mg BID vs placebo at week 16. Deucravacitinib was well tolerated, and AEs were consistent with the known safety profile. These data support further evaluation of deucravacitinib for the treatment of cutaneous manifestations of lupus, including in patients with SLE in the ongoing phase 3 POETYK SLE trials (NCT05617677, NCT05620407).
REFERENCES: [1] Morand E, et al. Arthritis Rheumatol 2023;75:242–252.
[2] Arriens C, et al. Oral presentation at ACR Convergence 2023; November 10-15, 2023; San Diego, CA. Abstract 2489.
Acknowledgements: We thank the patients and families who made this study possible, as well as the clinical teams that participated. The study was supported by Bristol Myers Squibb. All authors contributed to and approved the abstract; professional medical writing and editorial assistance was provided by Christine Billecke, PhD, of Nucleus Global, funded by Bristol Myers Squibb.
Disclosure of Interests: Joseph F Merola AbbVie, Amgen, AstraZeneca, Biogen, Boehringer Ingelheim, Bristol Myers Squibb, Dermavant, Eli Lilly, Incyte, Janssen, Leo Pharma, MoonLake, Novartis, Pfizer, Sanofi-Regeneron, Sun Pharma, and UCB, Alice B. Gottlieb Amgen, Eli Lilly, Highlight Therapeutics, Janssen, Novartis, Sanofi, Sun Pharma, Takeda, Teva, UCB, and XBiotech (stock options for RA), Bristol Myers Squibb, Janssen, MoonLake, and UCB Pharma (all paid to Mount Sinai School of Medicine), Cynthia Aranow GSK, Bristol Myers Squibb, Synthekine, Merck, Alumis, and AstraZeneca, François Chasset AstraZeneca, GSK, Amgen, Biogen, Bristol Myers Squibb, and Novartis, AstraZeneca, Biogen, Bristol Myers Squibb, GSK, Celgene, Principia Bio, Lilly, Horizon Therapeutics, Kyowa Kirin, and Novartis, AstraZeneca, Bristol Myers Squibb, and GSK, Jörg Wenzel medac, Biogen, Novartis, LEO, Kyowa Kirin, Janssen, and Sanofi, Actelion, AstraZeneca, Bayer, Biogen, Bristol Myers Squibb, GSK, Incyte, Janssen, and Merck, GSK, Incyte, Spirig, AstraZeneca, Bristol Myers Squibb, Almirall, Novartis, Merck/Serono, Roche, Pfizer, ArrayBio, LEO, and GSK, David Fiorentino Annexon, Arsenal, Bristol Myers Squibb, Biogen, Evommune, Johnson and Johnson, Paragon, Pfizer, ACELYRIN, argenx, and UCB, EMD/Serono, Priovant, argenx, Kyverna, and IgM, Cristina Arriens AstraZeneca and Aurinia, AstraZeneca, Aurinia, Bristol Myers Squibb, Cabaletta, GSK, Health & Wellness Partners, Kezar, Synthekine, and UCB, AstraZeneca and Bristol Myers Squibb, Benjamin Chong CESAS Medical, MAPI Trust (royalties), Amgen, AstraZeneca, Biogen, Bristol Myers Squibb, EMD Serono, and Lupus Research Alliance, Annegret Kuhn Almirall/Hermal, Bristol Myers Squibb, and EMD Serono, Ronald F. van Vollenhoven AbbVie, Galapagos, Janssen, Pfizer, and UCB, AbbVie, AstraZeneca, Biogen, Biotest, Celgene, Galapagos, Gilead, Janssen, Pfizer, Servier, and UCB, Bristol Myers Squibb, Eli Lilly, GSK, Roche, and UCB, Shimon Korish Bristol Myers Squibb, Bristol Myers Squibb, Nikolay Delev Bristol Myers Squibb, Bristol Myers Squibb, Richard Meier Bristol Myers Squibb, Bristol Myers Squibb, Rachana Agrawal Bristol Myers Squibb, Bristol Myers Squibb, Thomas Lehman Bristol Myers Squibb, Bristol Myers Squibb, Brandon Johnson Bristol Myers Squibb, Bristol Myers Squibb, Brandon Becker Bristol Myers Squibb, Bristol Myers Squibb, Jiyoon Choi Bristol Myers Squibb and Johnson & Johnson, Bristol Myers Squibb, Coburn Hobar Bristol Myers Squibb, Bristol Myers Squibb, Victoria P. Werth CESAS Medical, MAPI Trust (royalties), Janssen, Lilly, Pfizer, Biogen, Bristol Myers Squibb, Gilead, Amgen, Nektar, EMD Serono, CSL Behring, Crisalis, Viela Bio, argenx, Kyowa Kirin, Regeneron, AstraZeneca, AbbVie, GSK, Cugene, UCB, Rome Pharmaceuticals, Horizon, Merck, Sanofi, Calyx, Cabaletta Bio, Nuvig Pharmaceuticals, Takeda, Immunovant, AnaptysBio, Evommune, Innovaderm, Alpine Immune Sciences, Caribou, Xencor, and Ventus, Pfizer, Biogen, Gilead, Corbus Pharmaceuticals, AstraZeneca, Amgen, Regeneron, CSL Behring, Bristol Myers Squibb, Horizon, Rome Pharmaceuticals, Priovant, Ventus, and Viela.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (