

Background: For the treatment of radiographic axial spondyloarthritis (r-axSpA), the Assessment of SpondyloArthritis International Society and European Alliance of Associations for Rheumatology (ASAS-EULAR) recommendations suggest the use of Tumour Necrosis Factor inhibitors (TNFi), Interleukin-17 inhibitors (IL-17i), or Janus kinase inhibitors (JAKi) for patients with persistently high disease activity despite conventional treatments. Current practice is to start with TNFi or IL-17i [1]. Ixekizumab (IXE) is a humanized immunoglobulin G subclass 4 (IgG4) monoclonal antibody (MAb) that neutralizes the cytokine interleukin-17A and has previously demonstrated efficacy in biologic-naïve and biologic-experienced patients with r-axSpA with and without elevated C-reactive protein (CRP) or evidence of spinal inflammation on magnetic resonance imaging (MRI) [2]. However, these analyses were in some cases limited by small sample sizes, and it is beneficial to demonstrate efficacy in a wider and more diverse patient population. CRP level in axSpA patients is a generally accepted indicator of inflammation and is also considered to be a predictor of clinical response to TNFi [3]. Currently, the impact of CRP on b/tsDMARD response is highlighted by the ASAS-EULAR research agenda.
Objectives: To evaluate the efficacy of IXE in the phase III r-axSpA clinical development program through a pooled analysis of biologic-naïve patients stratified by CRP level.
Methods: Biologic-naïve patients from the COAST-V (NCT02696785, n = 168) [2] international trial and a study of Chinese patients (NCT04285229, n = 130) [4] with r-axSpA were pooled for analysis. These phase III, multicentre, double-blind studies randomly assigned patients to receive either PBO or IXE every 4 weeks (Q4W) from baseline to week 16 (W16) stratified by baseline CRP level, defined as normal (CRP ≤5 mg/L) or elevated (CRP >5 mg/L). The Assessment of SpondyloArthritis international Society 40 (ASAS40) response criteria and its individual components, achievement of Axial Spondyloarthritis Disease Activity Score low disease activity (ASDAS LDA) state, and a 50% improvement in the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI 50) were assessed as efficacy measures after 16 weeks of treatment. An alpha threshold of 0.05 was used for this post hoc analysis and no multiplicity testing adjustment was performed.
Results: Baseline disease burden as measured by ASAS response individual components, ASDAS-CRP, and BASDAI did not differ significantly between patients with normal and elevated CRP (Table 1). Patients treated with IXE showed a larger mean change from baseline (CFB) at W16 in ASAS40 response individual components compared with PBO for both normal and elevated CRP groups (Figure 1A and Figure 1B). A higher percent of patients treated with IXE also achieved ASAS40 for both groups: 45.5% (IXE) and 17.4% (PBO) for patients with normal CRP vs. 47.1% (IXE) and 12.9% (PBO) for patients with elevated CRP (Figure 1C). Similarly, a higher percentage of IXE-treated patients achieved ASDAS LDA at W16 compared with PBO for both groups, with 58.2% (IXE) and 23.9% (PBO) for patients with normal CRP vs. 44.8% (IXE) and 7.1% (PBO) for patients with elevated CRP (Figure 1D). Comparable efficacy was demonstrated by patients achieving BASDAI 50 at W16, regardless of CRP level: 41.8% (IXE) and 15.2% (PBO) for patients with normal CRP vs. 43.7% (IXE) and 14.9% (PBO) for patients with elevated CRP (Figure 1E).
Conclusion: These data indicate that IXE provides substantial benefits in the treatment of patients with r-axSpA with both normal and elevated baseline CRP levels.
REFERENCES: [1] Ramiro S, et al. ASAS-EULAR recommendations for the management of axial spondyloarthritis: 2022 update. Ann Rheum Dis. 2023;82(1):19-34.
[2] Maksymowych WP, et al. Ixekizumab in radiographic axial spondyloarthritis with and without elevated C-reactive protein or positive magnetic resonance imaging. Rheumatology (Oxford). 2022;61(11):4324-34.
[3] Landewe R, et al. A single determination of C-reactive protein does not suffice to declare a patient with a diagnosis of axial spondyloarthritis ‘CRP-negative’. Arthritis Res Ther. 2018;20(1):209.
[4] Xue Y, et al. Ixekizumab for Active Radiographic Axial Spondyloarthritis in Chinese Patients: 16- and 52-Week Results from a Phase III, Randomized, Double-Blind, Placebo-Controlled Study. BioDrugs. 2024;38(1):145-56.
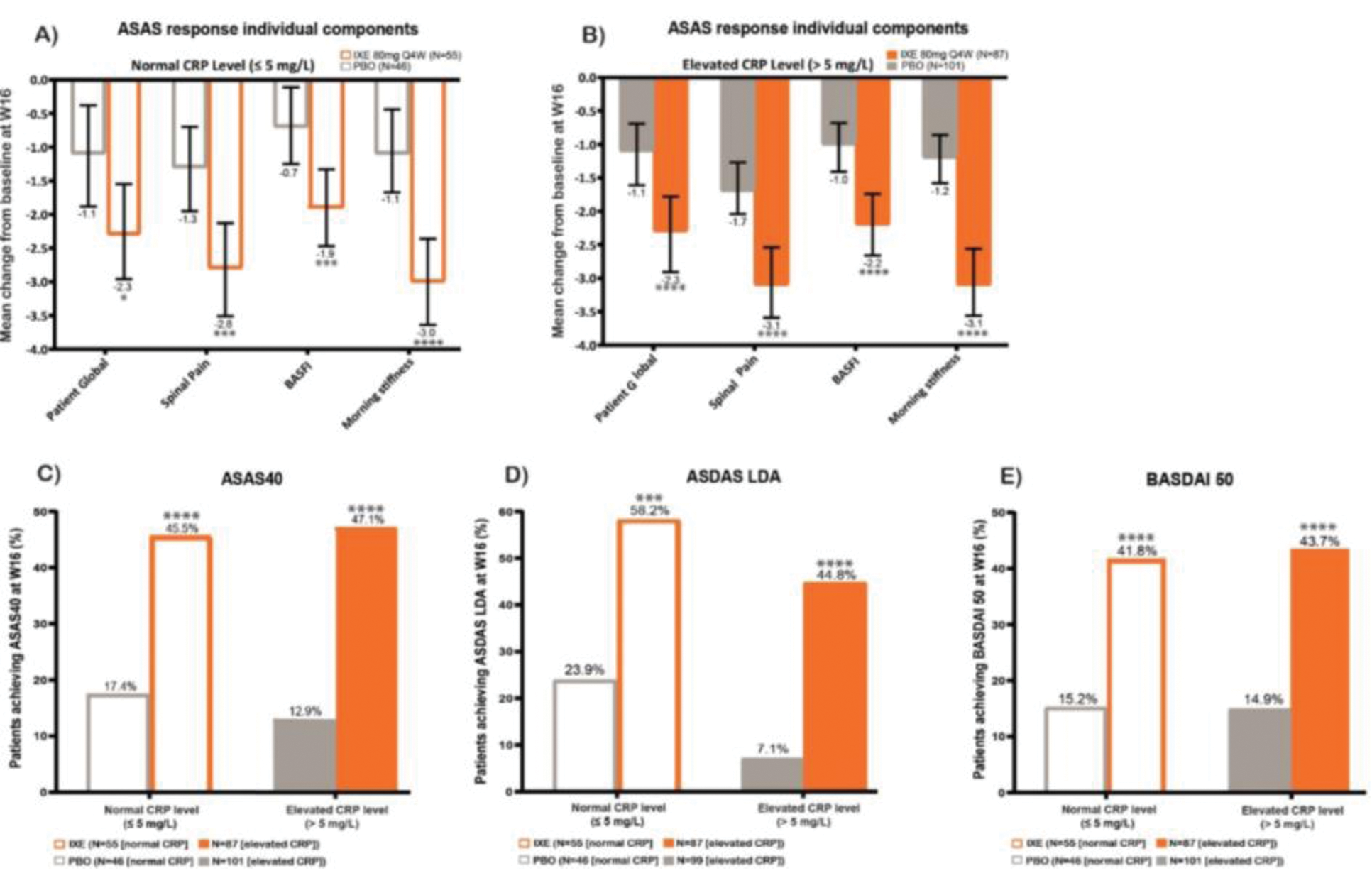
Table 1. Baseline characteristics of biologic-naïve patients from pooled phase III trials.
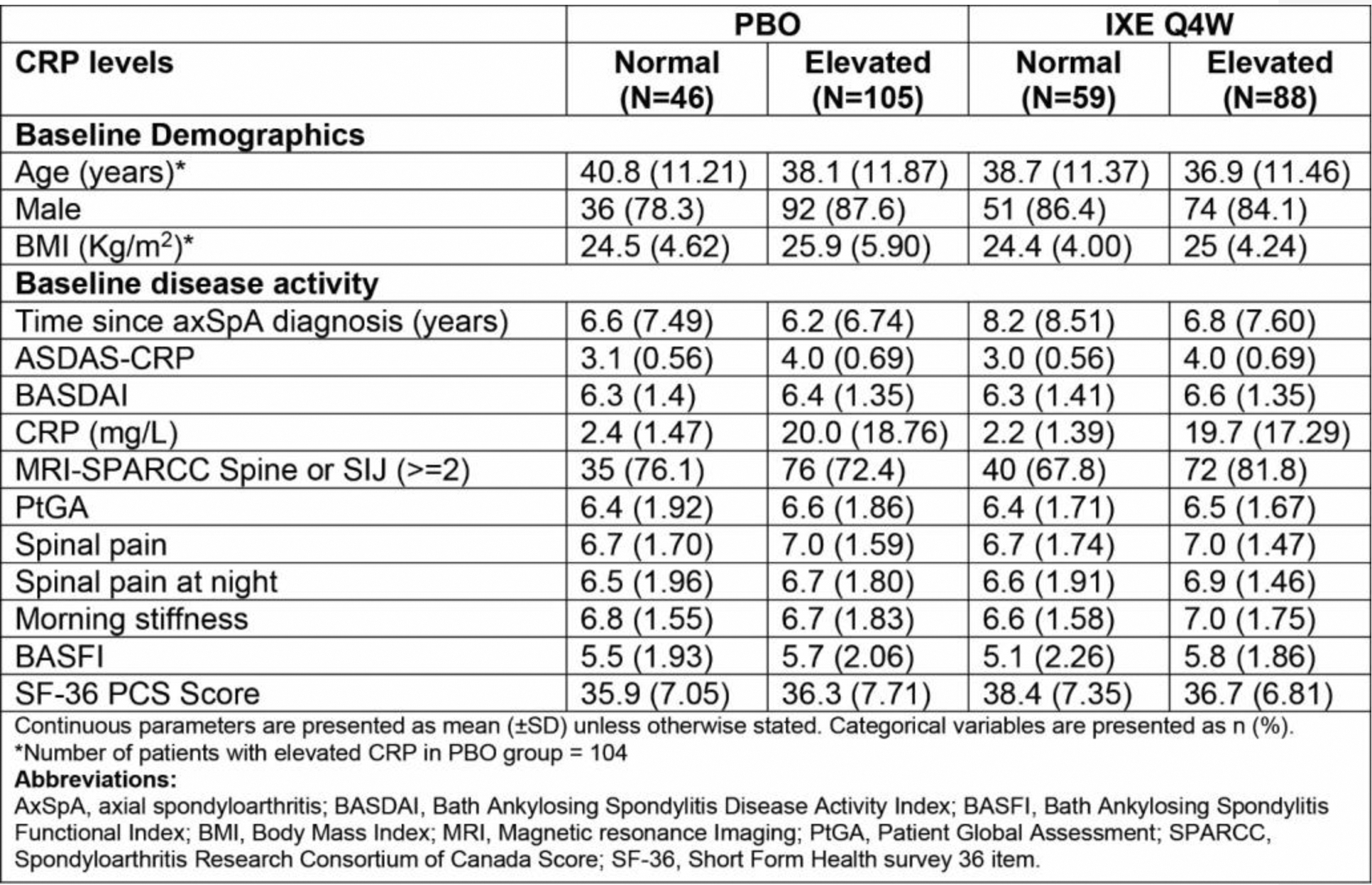
Efficacy outcomes for patients from pooled clinical trials.
A ) Mean CFB of ASAS response individual components at week 16 for patients with normal CRP. B ) Mean CFB of ASAS response individual components at week 16 for patients with elevated CRP. C ) ASAS 40 response (%) at W16 of patients receiving IXE vs. PBO stratified by CRP level. D ) Percent of patients achieving ASDAS low disease activity at W16 receiving IXE vs. PBO stratified by CRP level. E ) Percent of patients achieving BASDAI 50 at W16 receiving IXE vs. PBO stratified by CRP. * = p ≤ 0.05 ** = p ≤ 0.01 *** = p ≤ 0.001 **** = p ≤ 0.0001.
Acknowledgements: The authors would like to thank Shane Whelan for writing and editorial support.
Disclosure of Interests: Raj Sengupta Speakers bureau: AbbVie, UCB, Novartis, Eli Lilly and Company, Pfizer, Grant/research support from: Novartis, UCB, Xenofon Baraliakos Speakers bureau: AbbVie, Alphasigma, Amgen, BMS, Cesas, Celltrion, Galapagos, Janssen, Eli Lilly and Company, Moonlake, Novartis, Pfizer, Roche, Sandoz, Springer, Stada, Takeda, UCB, Zuellig, Consultant of: AbbVie, Alphasigma, Amgen, BMS, Cesas, Celltrion, Galapagos, Janssen, Eli Lilly and Company, Moonlake, Novartis, Pfizer, Roche, Sandoz, Springer, Stada, Takeda, UCB, Zuellig, Grant/research support from: Novartis, AbbVie, Celltrion, Janssen, Moonlake, Pedro Machado Speakers bureau: AbbVie, BMS, Celgene, Eli Lilly and Company, Galapagos/Alfasigma, Janssen, MSD, Novartis, Orphazyme, Pfizer, Roche, UCB, Consultant of: AbbVie, BMS, Celgene, Eli Lilly and Company, Galapagos/Alfasigma, Janssen, MSD, Novartis, Orphazyme, Pfizer, Roche, UCB, Philippe Goupille Speakers bureau: AbbVie, Amgen, BMS, Celltrion, Chugai, Janssen, Eli Lilly and Company, MSD, Novartis, UCB, Consultant of: AbbVie, Janssen, Eli Lilly and Company, MSD, Novartis, UCB, Mohamed Sheesh Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Khai Jing Ng Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Mizan Alam Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Rebecca Bolce Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Yan Yan Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Yu Xue: None declared, Martin Rudwaleit Speakers bureau: AbbVie, AstraZeneca, Janssen, Eli Lilly and Company, Novartis, UCB, Consultant of: AbbVie, AstraZeneca, Janssen, Eli Lilly and Company, Novartis, UCB, Grant/research support from: Janssen, UCB, Victoria Navarro-Compán Speakers bureau: AbbVie, Fresenius Kabi, Janssen, Eli Lilly and Company, Novartis, Pfizer, UCB, Consultant of: AbbVie, Alfasigma, Galapagos, Eli Lilly and Company, Novartis, Pfizer, UCB, Grant/research support from: Novartis, ASAS.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (