

Background: Currently known autoantibodies in Systemic Lupus Erythematosus (SLE) either lack specificity or present low sensitivity (e.g., anti-dsDNA and anti-Sm autoantibodies). At the same time, the antigen specificity for several autoantibodies that are present in SLE patients has yet to be determined. In a study involving patients with different autoimmune diseases and healthy controls, IgG and IgA anti-LIN28A antibodies were identified and validated as autoantibodies with a high specificity and sensitivity for SLE [1].
Objectives: To assess the presence, prevalence, and clinical correlates of IgG and IgA anti-LIN28A in active lupus nephritis (LN).
Methods: Adult patients with a biopsy-proven LN were enrolled between 2017 and 2023 from Cliniques universitaires San-Luc in Brussels, Belgium, and Padua University Hospital in Padua, Italy. Patients were regularly followed up; clinical and laboratory features were collected in a longitudinal fashion. A diagnostic kidney biopsy was performed at baseline. A subgroup of patients underwent a per-protocol repeat kidney biopsy at 12 months together with a second blood withdrawal. Patients’ serum samples were analysed using KREX technology-based microarrays (i-Ome Discovery; Sengenics) and screened for circulating IgG and IgA autoantibodies against 1609 proteins. Log2-transformed mean net signal intensities of IgG and IgA anti-LIN28A were median-normalised and batch-corrected together with measurements from 39 serum samples from healthy controls (HC) from the PRECISESADS clinical consortium [2]. A positivity cut-off for IgG and IgA anti-LIN28A was defined as median plus two interquartile ranges (IQRs) of the distribution levels of the antibodies in HC. Associations between anti-LIN28A levels and clinical and histological features were assessed at different time points. Data were analysed and compared using one-way ANOVA and t-test for continuous normally-distributed variables or chi-squared and Mann-Whitney U tests for non-parametric variables. Spearman’s rank correlation (r) was used to assess correlations between non-normally distributed variables.
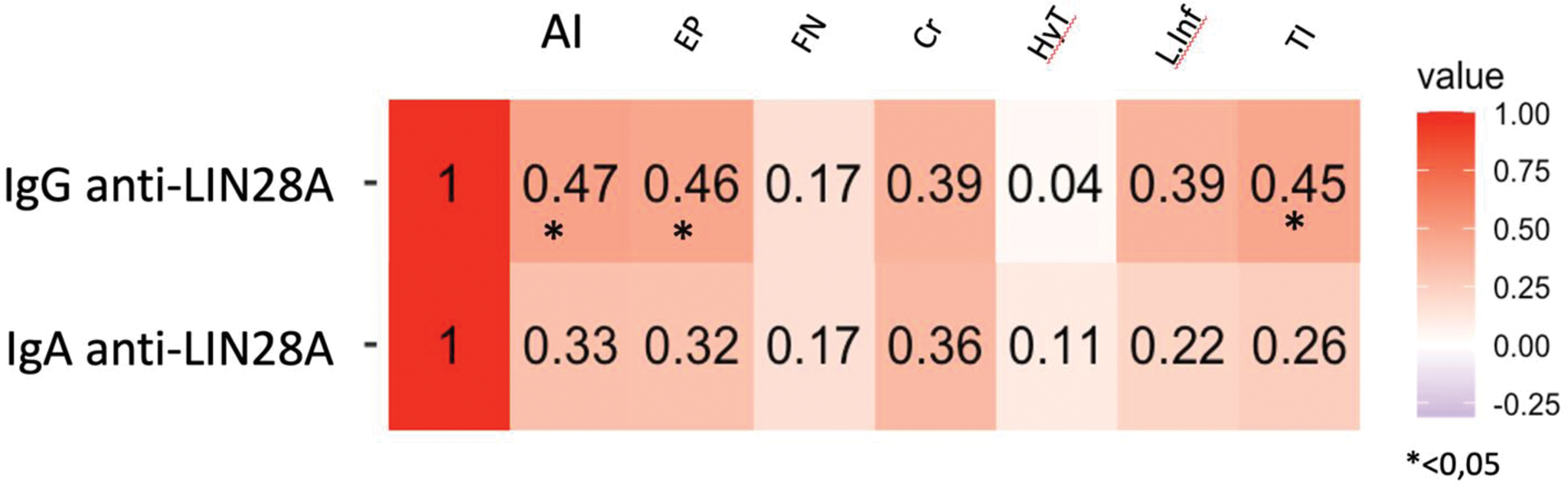
Results: Thirty-six patients with active, biopsy-proven, LN were enrolled (Table 1). At baseline, levels of IgG and IgA anti-LIN28A were significantly elevated in LN patients compared to HC (p<0.0005). Among LN patients, 87.0% and 77.8% were positive for IgG and IgA anti-LIN28A, respectively. Patients negative for the IgG isotype had significantly lower NIH Activity Index (AI) scores at the diagnostic kidney biopsy (p<0.005). There was a correlation between IgG anti-LIN28A levels and total NIH AI scores (r=0.47, p=0.011), mainly driven by endocapillary proliferation (r=0.46, p0.0219) and tubulointerstitial mononuclear cell infiltration (r=0.45, p=0.022) (Figure 1). Patients displaying cellular crescents at the diagnostic biopsy showed significantly higher levels of IgG (p=0.0145) and IgA (p=0.04) anti-LIN28A. After 12 months of treatment, autoantibody levels were significantly reduced (p<0.001 for both isotypes). Among 18 patients who underwent a per-protocol repeat kidney biopsy, 2 patients with persistent histological inflammation (NIH AI scores >0) displayed a milder decrease in IgG anti-LIN28A levels (p<0.05). Moreover, patients showing EULAR renal response (proteinuria <0.5 g/day and stable/improved eGFR) [3] showed a tendency towards a more prominent decline in IgG anti-LIN28A compared to that seen in non-responders, but this difference did not reach statistical significance (p=0.07). No correlation was seen between changes in IgA anti-LIN28A levels and histological or clinical response to treatment for LN.
Conclusion: IgG and IgA anti-LIN28A antibodies are markedly elevated in active LN patients. IgG anti-LIN28A showed a significant association with inflammation at the histological level, especially with endocapillary proliferation and tubulointerstitial inflammatory infiltrates. Moreover, patients achieving histological remission in per-protocol repeat kidney biopsy (NIH AI=0) displayed a more prominent decrease in IgG anti-LIN28A levels compared with LN patients with persistent histological inflammation in the repeat biopsy (NIH AI>0). Our data point to a potential role for IgG anti-LIN28A antibodies as diagnostic and prognostic markers of LN.
Table 1. Characteristics of patients and healthy controls.
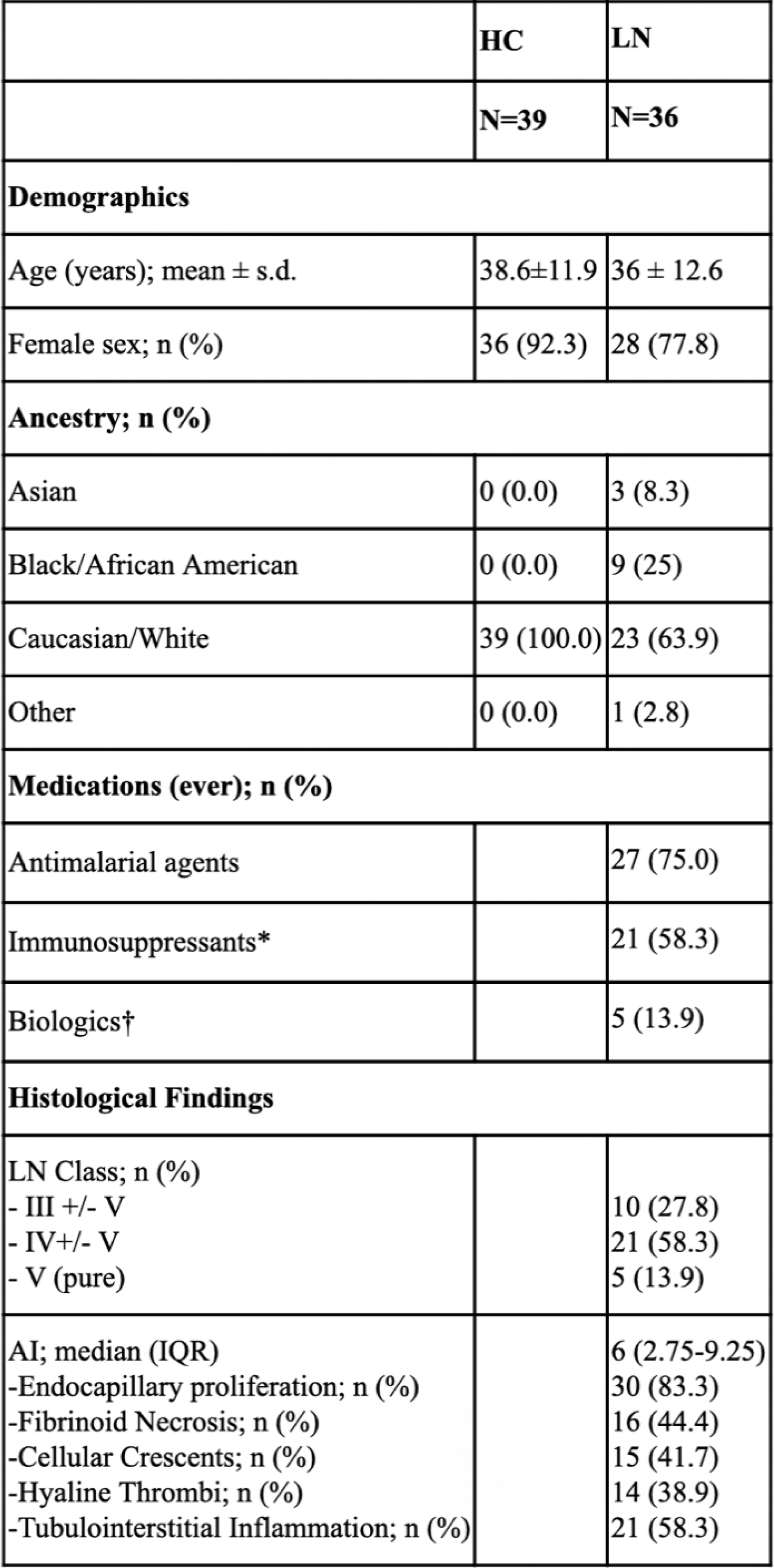
* Azathioprine, calcineurin inhibitors, leflunomide, methotrexate, mycophenolic acid, or sulfasalazine, cyclophosphamide.
† rituximab or belimumab.
HC: healthy controls; IQR: interquartile range; s.d.: standard deviation.
Correlations between IgG and IgA anti-LIN28A levels and NIH Activity Index total and item scores.
Heatmap showing Spearman’s rank correlation coefficients (r) of IgG and IgA anti-LIN28A levels with NIH AI total and item scores.
AI, Activity Index; Cr, cellular and fibro-cellular crescents; EP, endocapillary proliferation; FN, fibrinoid necrosis; HyT, hyaline thrombi; L.inf, lymphocytic infiltrate; r, Spearman’s rank correlation coefficient; TI, tubulointerstitial mononuclear cell infiltration.
REFERENCES: [1] Parodis I, Nikolopoulos D, Lindblom J, et al. IgG and IgA anti-LIN28A Outperform Anti-dsDNA and anti-Sm in Distinguishing SLE from Health and Other Autoimmune Diseases [abstract]. Arthritis Rheumatol. 2024; 76 (suppl 9).
[2] Barturen G, Babaei S, Català‐Moll F, et al. Integrative Analysis Reveals a Molecular Stratification of Systemic Autoimmune Diseases. Arthritis & Rheumatology 2021; 73: 1073–1085.
[3] Fanouriakis A, Kostopoulou M, Alunno A, et al. 2019 update of the EULAR recommendations for the management of systemic lupus erythematosus. Ann Rheum Dis 2019; 78: 736–745.
Acknowledgements: PRECISESADS Clinical Consortium.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (