

Background: The Schirmer test is one of the established diagnostic tests for suspected Sjögren’s Disease (SjD) and is used to objectify ocular sicca. It can be divided into Schirmer 1 (S1) and Schirmer 2 (S2) tests. In both tests, a filter paper strip is inserted into the lower eyelid and the length of the soaked area on the filter paper strip is measured after 5 minutes. This test can be performed with (S1) or without (S2) local anaesthetic. The S1 test detects all tear production and the S2 test records primarily the basal secretion without testing the reflex production caused by the foreign body sensation when the filter paper strip is inserted.
Objectives: The aim of the study was to analyze the predictive power of the two tests in order to make a recommendation as to which of the two tests, in our opinion and by adjusting the cut-off value, could be optimal in everyday clinical practice.
Methods: For this purpose, 200 eyes of patients with SjD and 104 eyes of age- and sex-matched control subjects were examined. The S1 and S2 tests were standardized, with an appropriate time window between the two tests. In addition, each participant completed a questionnaire with information on clinical symptoms, and we supplemented this data with information from the patients’ files, such as antibody status.
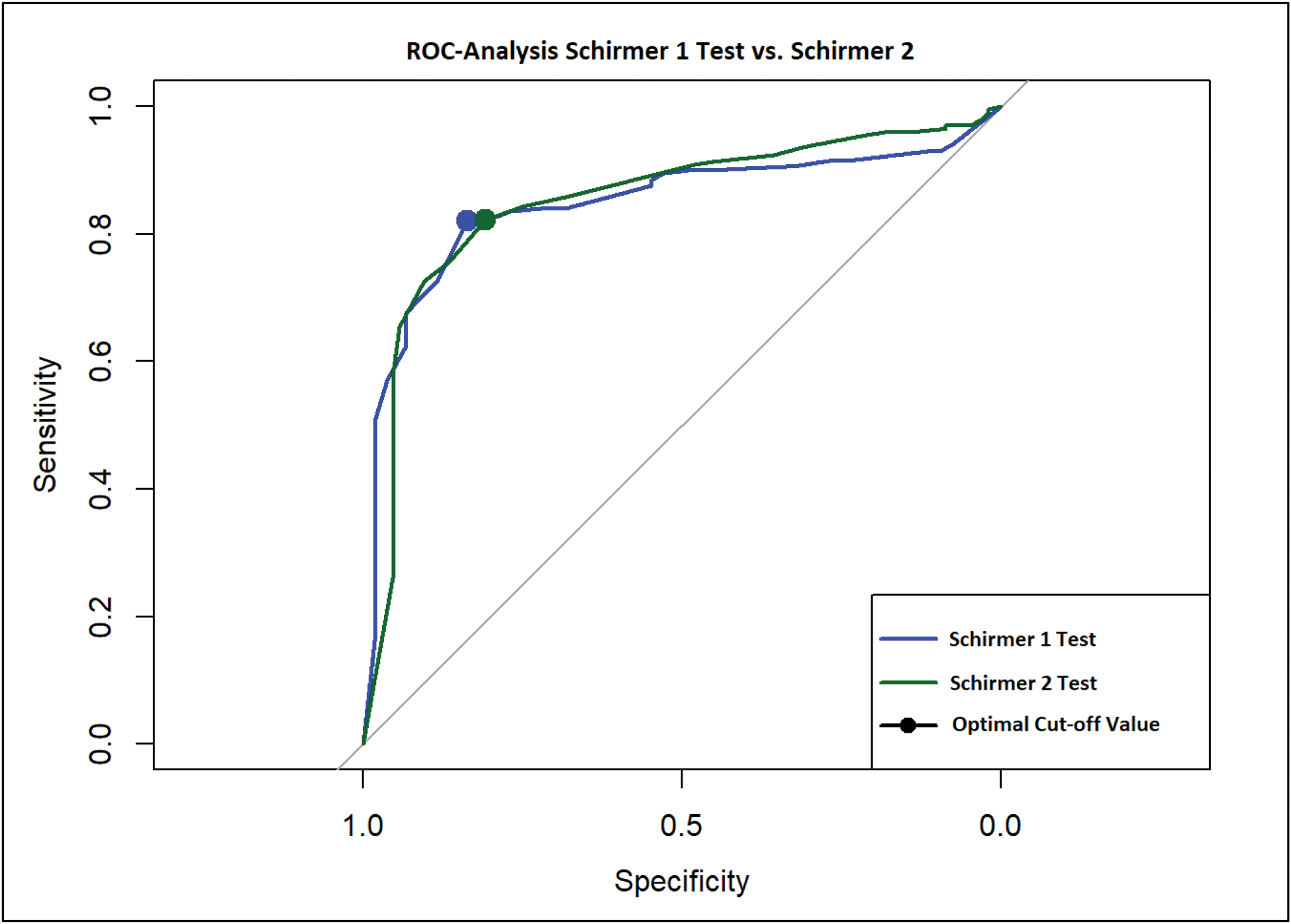
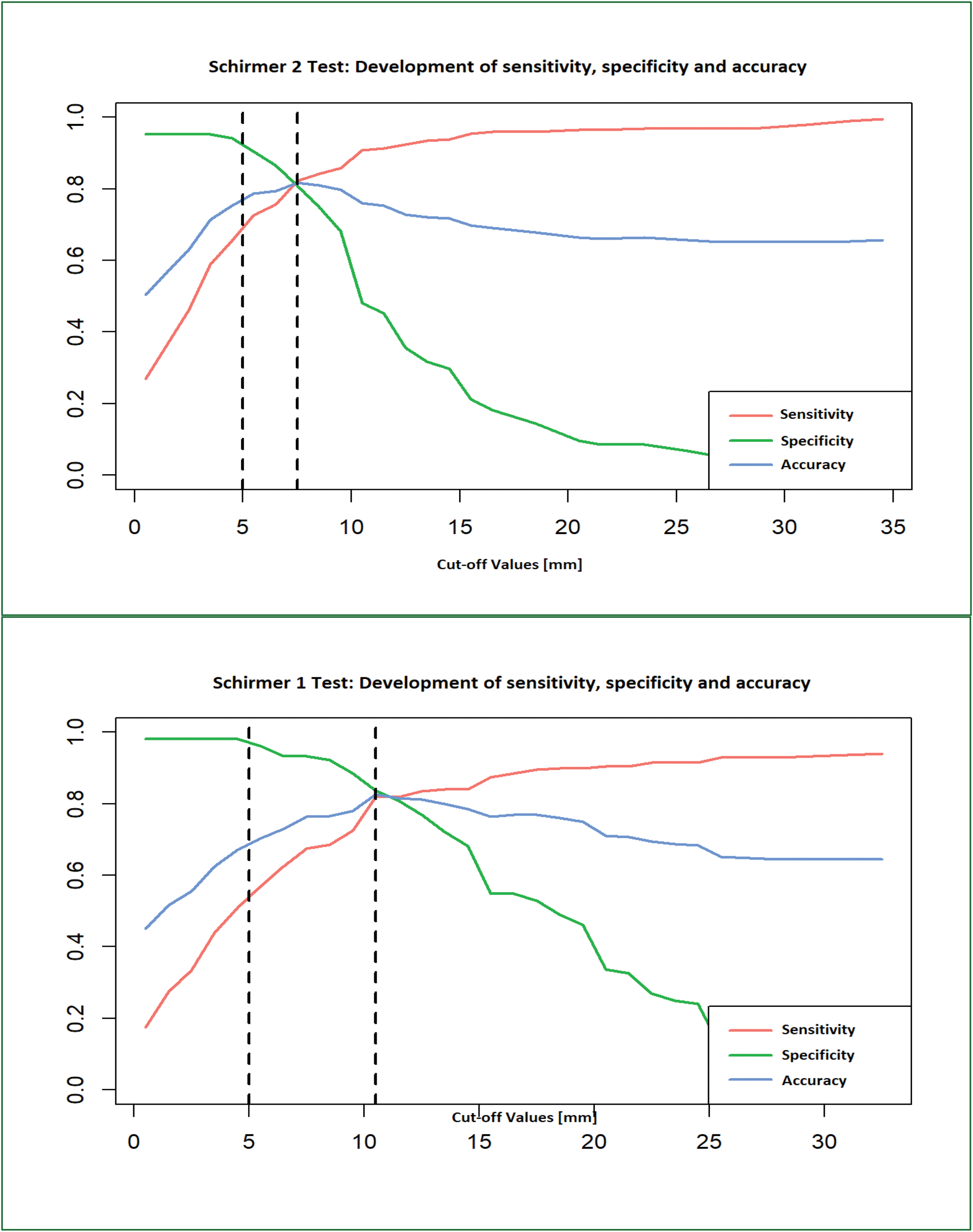
Results: The S1 test was pathological in 114/200 (57%) eyes in the SjD cohort and in 4/104 (4%) control eyes. The S2 test showed pathological findings in 143/200 (71.5%) eyes in the SjD cohort and in 10/104 (9.6%) control eyes. In the overall cohort, 114/304 eyes (37.5%) had pathological findings on both Schirmer tests. In addition, 39 eyes were identified that did not have a pathological S1 test but had an abnormal S2 test. The S1 test had a sensitivity of 96.2% and a specificity of 57% in our cohort. The S2 test had a specificity of 72.6% and a sensitivity of 90.4%. In a direct comparison and taking into account the prevalence of ocular dryness in SjD patients reported in the literature to be 89% [1], a similar positive predictive value of 94.8% (S1) and 96.4% (S2) was found, whereas the negative predictive value was significantly different: 64.7% vs. 48.3%. The subsequent ROC analysis showed only a small difference in the development of sensitivity and specificity with different cut-off values for the two Schirmer tests (Figure 1). The cut-off with the best accuracy is shown for the S1 test at 10.5 mm [Sens=82.0%; Spec=83.7%]. For the S2 test, the optimum was found to be 7.5 mm [Sens=82.2%; Spec=80.8%]. In contrast, the standard cut-off of 5 mm has an accuracy of only 70.4% and 78.7% in our cohort (Figure 2).
Conclusion: The S2 test is more sensitive in detecting symptoms of ocular sicca in our cohort. However, as the ROC analysis shows only marginal differences, we cannot determine a significant superiority for the S2 test. Nevertheless, we could achieve better sensitivity with only a slight reduction in specificity if we marginally increase the cut-off value of the Schirmer 1 test.
Both tests have advantages and disadvantages: the use of local anaesthetic can lead to (rare) allergic reactions and patients show a reduced corneal reflex for a few hours, making corneal injury more likely. On the other hand, local anaesthesia can be used to examine patients who were previously unable to tolerate the insertion of the test paper strip. To summarize our results, we would consider the S2 test as a complementary test that could be used when the S1 test is borderline negative or when patients cannot tolerate it. Additionally, we would like to discuss a slight increase of the Cut-off value for the Schirmer 1 test.
REFERENCES: [1] Negrini S, Emmi G, Greco M, Borro M, Sardanelli F, Murdaca G, Indiveri F, Puppo F. Sjögren’s syndrome: a systemic autoimmune disease. Clin Exp Med. 2022 Feb;22(1):9-25. doi: 10.1007/s10238-021-00728-6. Epub 2021 Jun 7. PMID: 34100160; PMCID: PMC8863725.
ROC-Analysis of Schirmer 1 vs. Schirmer 2 test: The dot marks the optimal cut-off value.
Comparative illustration of the development of Sensitivity, Specificity and Accuracy in Schirmer 1 and Schirmer 2 test depending on the selected cut-off value: The first dashed line marks the standard cut-off value (5 mm) and the second dashed line marks the statistically optimal cut-off value, corresponding to the highest accuracy. The optimum Schirmer 1 cut-off is 10.5 mm, while the optimum Schirmer 2 cut-off is 7.5 mm.
Acknowledgements: NIL.
Disclosure of Interests: Nadine Zehrfeld: None declared, Gyde Tadsen: None declared, Sabrina Benz: None declared, Torsten Witte The Author received honoraria for lectures by Abbvie, BMS, Chugai, Galapagos, Janssen, Lilly, Pfizer, UCB and Roche. The Author received funding from the DFG (German Research Foundation) under Germany’s Excellence Strategy - EXC 2155 - project number 390874280. Katerina Hufendiek: None declared, Diana Ernst The Author received presentation honoraria from Abbvie, Amgen, BMS, Chugai, Cilag-Janssen, Galapagos, GSK, Medac, Lilly, Pfizer, Novartis, Roche. The Author participated in advisory boards for Abbvie, Galapagos, Amgen and Novartis. The Author received research grants by Novartis and Abbvie.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (