

Background: The standard treatment for polymyalgia rheumatica (PMR) is corticosteroid therapy. However, interleukin-6 receptor (IL-6R) inhibitor sarilumab is approved in the USA and some recommendations call for the use of either tocilizumab or sarilumab. Furthermore, the efficacy of several targeted therapies has been evaluated in PMR in recent years. The use of one (and in the future, multiple) targeted therapy(ies) raises several questions.
Objectives: Our objectives were: first, to identify biomarkers enabling the selection of the appropriate targeted therapy for a given patient and second, to identify biomarkers enabling the monitoring of disease activity (particularly in patients receiving IL-6R inhibitors, which significantly lower C-reactive protein [CRP] levels).
Methods: We conducted an ancillary study involving five clinical trials (four randomized, placebo-controlled trials and one open-label study) from two different countries, evaluating the efficacy of four different targeted therapies (tocilizumab in two studies, abatacept, baricitinib, and rituximab) in PMR. Additionally, we included 20 patients with PMR treated with corticosteroids and 20 healthy elderly subjects (HES). Serum samples were obtained prior to initiating treatment and three months after treatment initiation in the five clinical trials, at three months for corticosteroid-treated patients, and at a single time point for the HES group. Demographic, clinical, disease activity, and biological data were collected at the time of each sample. Based on our understanding of the disease and on previous results [1, 2], 24 markers (IL-6, sIL-6R, IL-1ra, PTX-3, YKL-40, Gal-3, Angpt 1&2, PDGF-AA, PDGF-BB, VEGF, VEGF-R2, CCL2, CCL5, CCL7, CXCL-10, MMP1, MMP3, OPN, TIMP-1, sCD25, sCD163, sPD-L1, FAP) were measured on 415 samples using the Luminex® platform. The data were analyzed using GraphPad software.
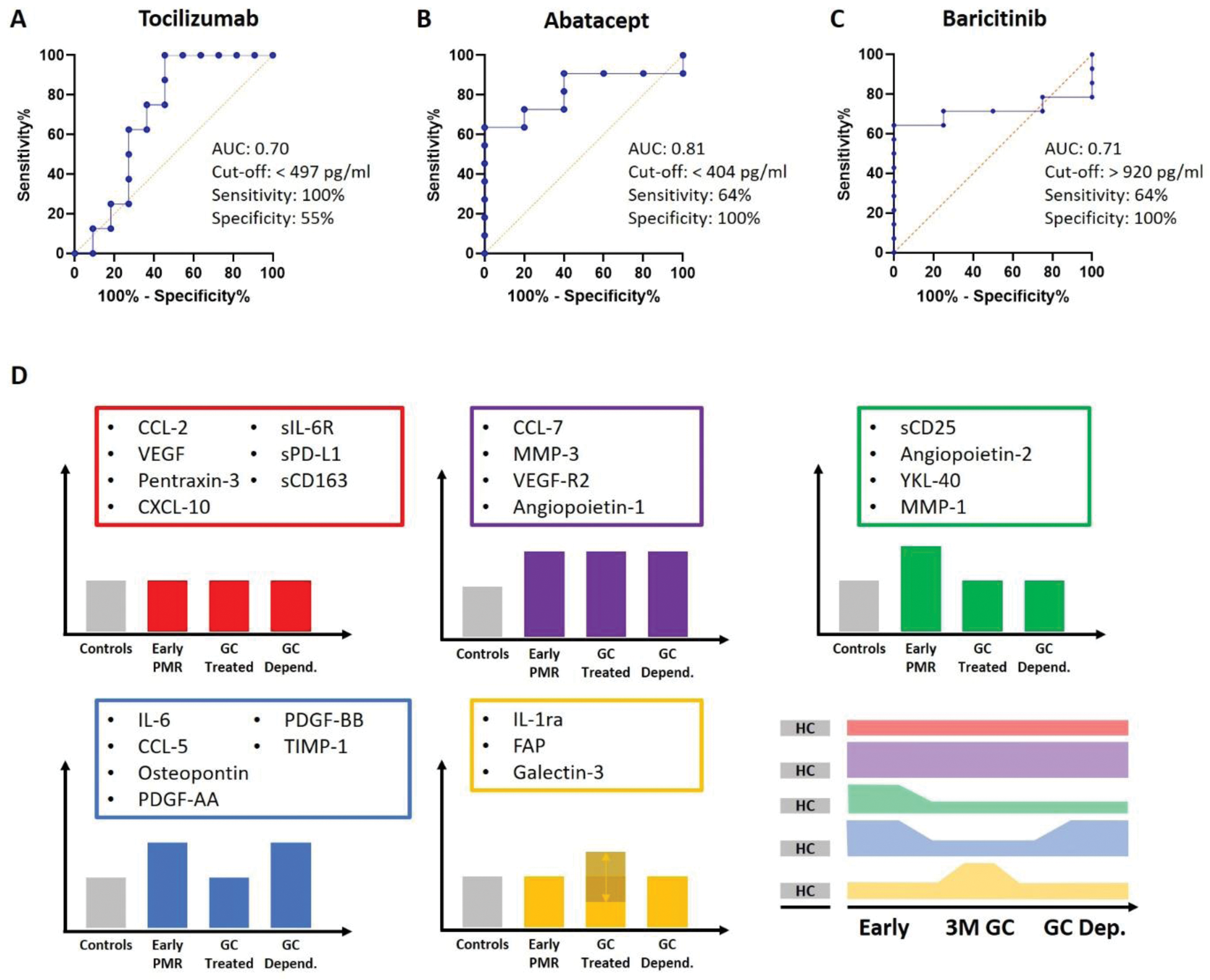
Results: The characteristics of the 227 patients and HES included in the study are presented in Table 1. The percentage of women was lower than expected in two studies. In three randomized, placebo-controlled trials, the patients had not received any glucocorticoids (GC) prior to initiating treatment. Initially, we assessed the external validity of our results by comparing them with published data from similar cohorts. We observed increases in interleukin-6 (p < 0.0001), angiopoietin-2 (p < 0.01), MMP-3 (p < 0.01), and YKL-40 (p < 0.0001) in patients with recent-onset PMR compared to healthy subjects, consistent with previous findings [1, 2]. The correlation between interleukin-6 levels measured by Luminex® and ELISA was very strong (r = 0.835; p < 0.0001). The biomarker sCD25 demonstrated good performance in predicting a favorable response to tocilizumab when the level was below 497 pg/mL, with an area under the curve (AUC) of 0.7 and 100% sensitivity (Figure 1A). A level below 404 pg/mL had an AUC of 0.81 and 100% sensitivity for predicting a favorable response to abatacept (Figure 1B) and a level over 920 pg/ml had an AUC of 0.71 and 100% specificity to predict a favorable response to baricitinib (Figure 1C). We identified five biomarker patterns based on the stage of the disease (Figure 1D). IL-6, CCL-5, osteopontin, PDGF-AA, PDGF-BB and TIMP-1 levels were higher in patients with an active disease compared to HEC and patients with an inactive disease under GC.
Conclusion: In this study, we identified a biomarker (sCD25) that may assist in selecting a targeted therapy and some candidates in monitoring disease activity. These findings will need to be confirmed in a validation cohort.
REFERENCES: [1] van Sleen Y, Boots AMH, Abdulahad W H, Bijzet J, Sandovici M, van der Geest KSM, et al. High angiopoietin-2 levels associate w ith arterial inflammation and long-term glucocorticoid requirement in polymyalgia rheumatica. Rheumatology. 10 juill 2019;kez261.
[2] van der Geest KSM, Abdulahad W H, Rutgers A, Horst G, Bijzet J, Arends S, et al. Serum markers associated w ith disease activity in giant cell arteritis and polymyalgia rheumatica. Rheumatology. août 2015;54(8):1397‑402.
Characteristics of the patients and controls included in the study.
| Characteristics | Healthy elderly subjects | GC | Tocilizumab** | Abatacept* | Baricitinib* | Rituximab* | Tocilizumab* |
|---|---|---|---|---|---|---|---|
| Number | 20 | 20 | 19 | 34 | 33 | 24 | 77 |
| Age (years ) | 74
| 71
| 67
| 71
| 70
| 65
| 67
|
| Women | 14
| 12
| 7
| 22
| 21
| 8
| 52
|
| Height (cm ) | 169
| 167
| 168
| 163
| 164
| 177
| 162
|
| Weight (kg ) | 74
| 74
| 81
| 68
| 68
| 79
| 73
|
| Body mass index | 26
| 26
| 28
| 26
| 25
| 25
| 27
|
| Symptom duration (months ) | NA | 4
| 3
| 8
| 2
| 3
| 19
|
| GC at baseline (mg/j ) | NA | 12
| 0
| 0
| 0
| 14
| 10
|
| PMR-AS | NA | NA | 37
| 35
| 30
| 18
| 19
|
|
CRP
| 2
| 4
| 89
| 36
| 22
| 7
| 9
|
|
Methotrexate use
|
|
|
|
|
|
|
|
CRP: C-reactive protein, GC: glucocorticoids, PMR-AS: polymyalgia rheumatica activity score. *randomized controlled trials; ** open label trial.
Different patterns of biomarkers.
A-C Receiver operating characteristics curves for the response to tocilizumab (A), abatacept (B) or baricitinib (C) defined by achieving the primary outcome of the respective trial in early polymyalgia rheumatica. D Different patterns of biomarkers based on their level in controls (healthy elderly subjects), early untreated polymyalgia rheumatica (PMR) patients, PMR patients treated with glucocorticoids (GC) at 3 months and GC-dependent PMR patients.
Acknowledgements: NIL.
Disclosure of Interests: Guillermo Carvajal Alegria Abbvie, AlphaSigma, Biogen, BMS, Celltrion, Chugai, Fresenius-Kabi, Galapagos, Lilly, MSD, Novartis, Pfizer, Novartis, Yannick van Sleen: None declared, Alain Saraux: None declared, Divi Cornec: None declared, Kornelis van der Geest: None declared, Aatke van der Maas: None declared, Valerie Devauchelle-Pensec: None declared, Elisabeth Brouwer: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (