

Background: Axial spondyloarthritis (axSpA) is a chronic, immune-mediated, inflammatory disease, mainly affecting the sacroiliac joints (SIJ) and spine. Bimekizumab (BKZ) is a monoclonal IgG1 antibody that selectively inhibits interleukin (IL)-17F in addition to IL-17A. BKZ has demonstrated sustained efficacy and safety to Week 52 in patients across the full spectrum of axSpA in the phase 3 studies BE MOBILE 1 (non-radiographic [nr-]axSpA) and BE MOBILE 2 (radiographic [r-]axSpA) [1]. Achievement of remission is a treatment goal and may guide clinical decisions [2, 3]. There is currently no universally accepted definition of remission in axSpA for use in research and routine clinical practice [4]. Assessing remission using objective signs of inflammation (OSI) may provide a clearer picture of inflammatory disease activity without subjective criteria. Previous analyses have shown that higher proportions of patients receiving BKZ achieved remission based on OSI (MRI remission of the SIJ and spine, C-reactive protein [CRP] ≤5 mg/L and a swollen joint count [SJC] of 0) compared with Axial Spondyloarthritis Disease Activity Score <1.3 (ASDAS Inactive Disease [ID]) criteria in BE MOBILE 1 and 2 [5].
Objectives: To report achievement of a broader definition of OSI remission (using the previously used definition above, with the addition of absence of uveitis flares) in patients with axSpA compared with an established endpoint, ASDAS ID.
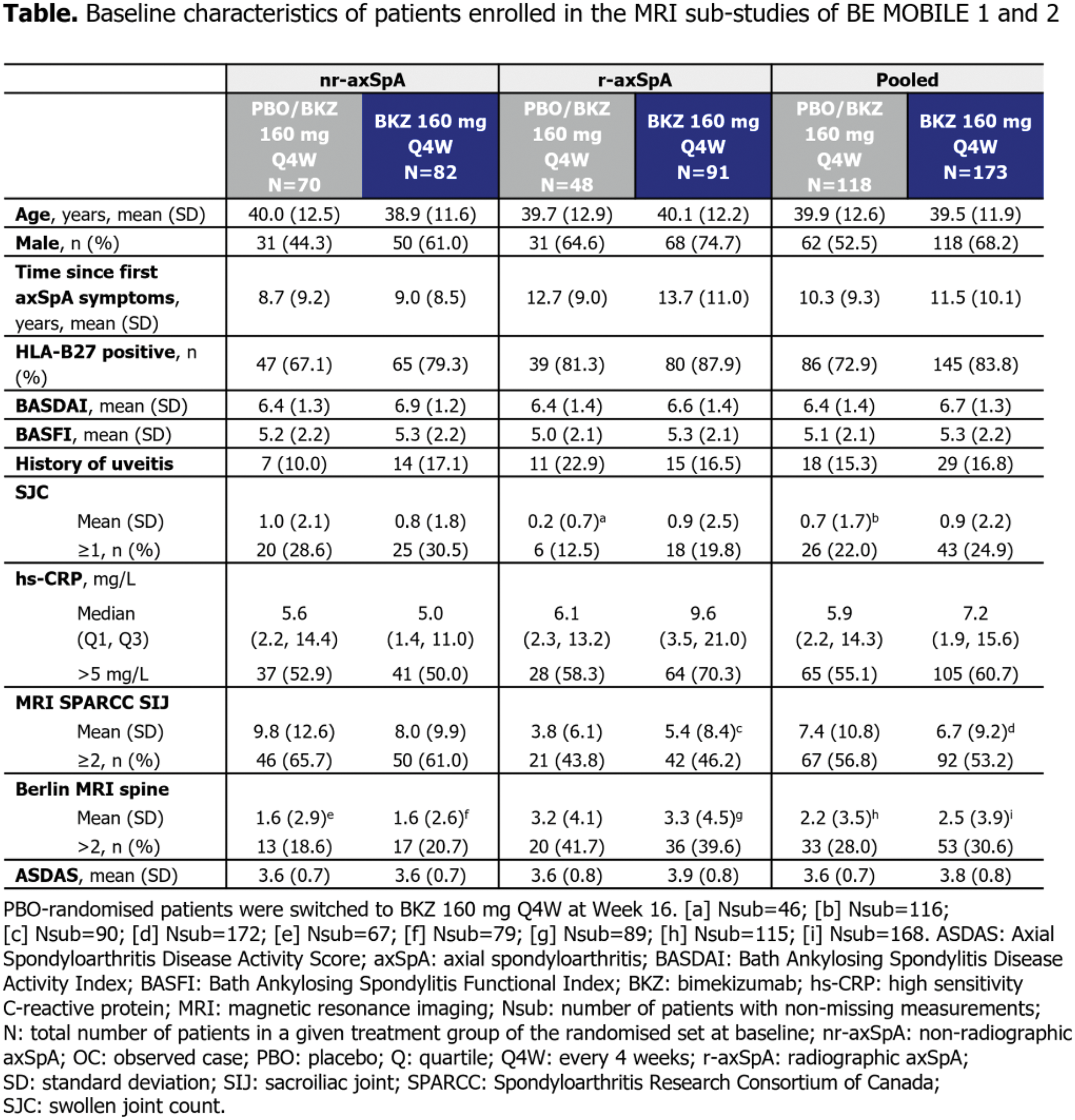
Methods: BE MOBILE 1 (NCT03928704) and BE MOBILE 2 (NCT03928743) comprised 16-week double-blind, placebo (PBO)-controlled periods followed by 36-week maintenance periods. Patients were randomised to subcutaneous BKZ 160 mg every 4 weeks (Q4W) or PBO, with all patients receiving BKZ from Week 16 onwards. Remission of OSI comprised MRI remission of the SIJ and spine (MRI Spondyloarthritis Research Consortium of Canada [SPARCC] SIJ score <2 and Berlin MRI spine score ≤2), CRP ≤5 mg/L, SJC of 0 and no uveitis flares from baseline to Week 52. The proportion of patients from the BE MOBILE 1 and 2 MRI sub-studies achieving these criteria was compared with those achieving ASDAS ID. No formal statistical analyses were conducted, and observed case (OC) data are reported.
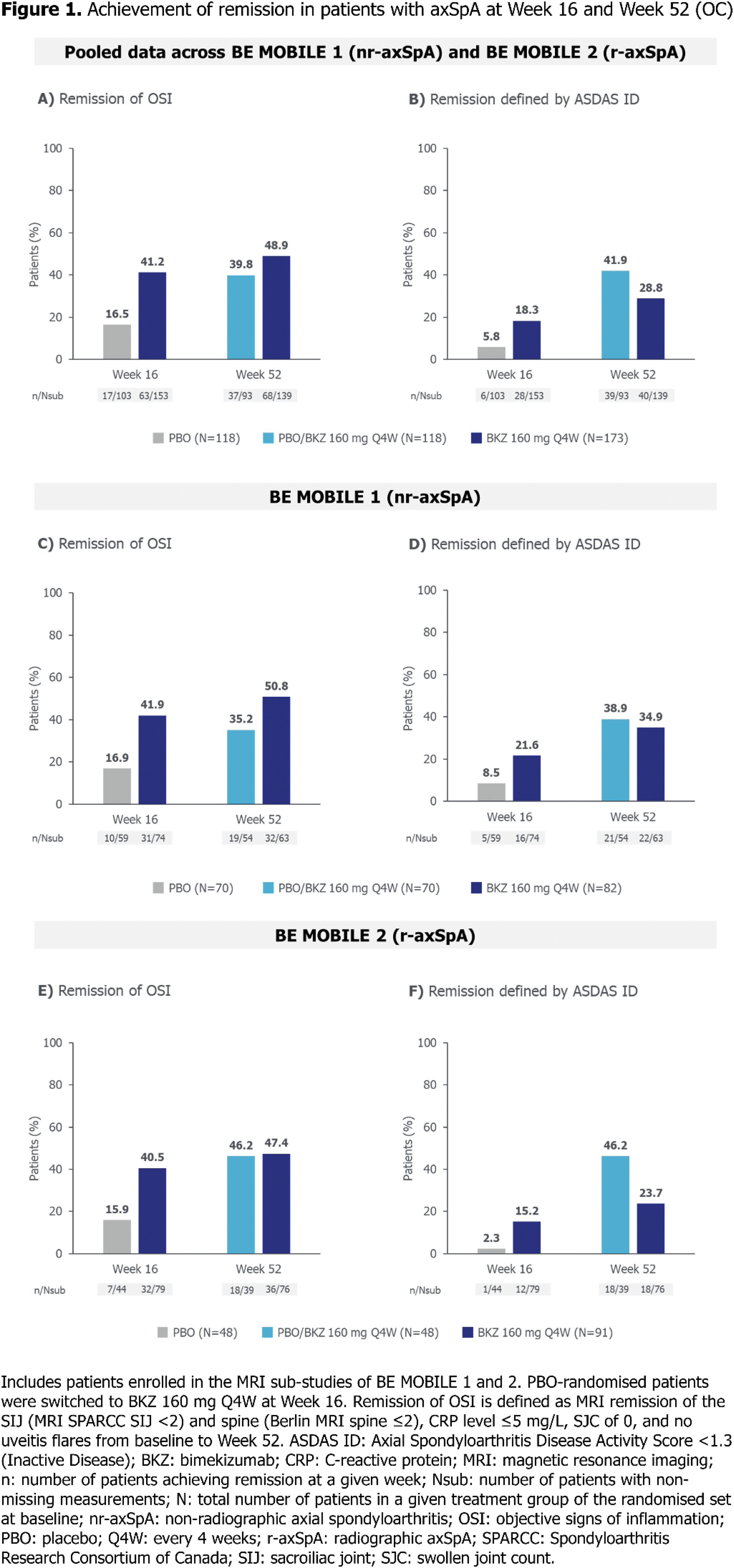
Results: Of 586 patients enrolled in BE MOBILE 1 and 2, 291 patients from their MRI sub-studies were included in this analysis. Levels of OSI at baseline were similar across treatment arms in patients with nr-axSpA and r-axSpA, respectively (Table 1). Overall, of pooled patients enrolled in the MRI sub-studies across BE MOBILE 1 and 2, 63/153 (41.2%) BKZ-randomised patients achieved remission of OSI at Week 16, compared with 28/153 (18.3%) achieving ASDAS ID. 17/103 (16.5%) PBO-randomised patients achieved remission of OSI at Week 16, compared with 6/103 (5.8%) achieving ASDAS ID. 68/139 (48.9%) BKZ-randomised patients achieved remission of OSI at Week 52, compared with 40/139 (28.8%) achieving ASDAS ID. Having switched to BKZ at Week 16, 37/93 (39.8%) PBO-randomised patients achieved remission of OSI at Week 52, compared with 39/93 (41.9%) achieving ASDAS ID (Figures 1A–1B). Separately, among patients with nr-axSpA, 31/74 (41.9%) BKZ-randomised patients achieved remission of OSI at Week 16, compared with 16/74 (21.6%) achieving ASDAS ID. 10/59 (16.9%) PBO-randomised patients achieved remission of OSI at Week 16, compared with 5/59 (8.5%) achieving ASDAS ID. 32/63 (50.8%) BKZ-randomised patients achieved remission of OSI at Week 52, compared with 22/63 (34.9%) achieving ASDAS ID. Having switched to BKZ at Week 16, 19/54 (35.2%) PBO-randomised patients achieved remission of OSI at Week 52, compared with 21/54 (38.9%) achieving ASDAS ID (Figures 1C–1D). In patients with r-axSpA, 32/79 (40.5%) BKZ-randomised patients achieved remission of OSI at Week 16, compared with 12/79 (15.2%) achieving ASDAS ID. Among PBO-randomised patients, 7/44 (15.9%) achieved remission of OSI at Week 16, compared with 1/44 (2.3%) achieving ASDAS ID. 36/76 (47.4%) BKZ-randomised patients achieved remission of OSI at Week 52, compared with 18/76 (23.7%) achieving ASDAS ID. Having switched to BKZ at Week 16, 18/39 (46.2%) PBO-randomised patients achieved remission of OSI at Week 52, compared with 18/39 (46.2%) achieving ASDAS ID (Figures 1E–1F).
Conclusion: A higher proportion of patients receiving BKZ achieved remission based on OSI compared with ASDAS ID criteria across the full disease spectrum of axSpA. This highlights the potential limitations of using ASDAS ID alone to assess treatment efficacy. These findings underscore the need for further research to improve endpoints in axSpA.
REFERENCES: [1] Baraliakos X. Ann Rheum Dis 2024;83:199–213.
[2] Smolen JS. Ann Rheum Dis 2018;77:3–17.
[3] Ramiro S. Ann Rheum Dis 2023;82:19–34.
[4] Baraliakos X. J Rheumatol 2018;45:153–7.
[5] Gensler LS. ACR 2024 [2362].
Acknowledgements: Funded by UCB. Medical writing support provided by Costello Medical and funded by UCB.
Disclosure of Interests: Martin Rudwaleit Speakers bureau from AbbVie, Boehringer Ingelheim, Chugai, Eli Lilly, Janssen, Novartis, Pfizer and UCB, Consultant of AbbVie, Eli Lilly, Novartis and UCB, Helena Marzo-Ortega Speaking honoraria from AbbVie, Amgen, Biogen, Eli Lilly, Janssen, MoonLake, Novartis, Pfizer, Takeda and UCB, Consultancy fees from AbbVie, Amgen, Biogen, Eli Lilly, Janssen, MoonLake, Novartis, Pfizer, Takeda and UCB, Research grants from Janssen, Novartis, Pfizer and UCB, Vanessa Taieb Shareholder of UCB, Employee of UCB, Diana Voiniciuc Contractor for UCB and employee of Veramed, Alexander Marten Employee of UCB, George Stojan Employee of UCB, Mindy Kim Employee of UCB, Lianne S Gensler Consulting fees from Acelyrin, Eli Lilly, Janssen, Novartis, Pfizer and UCB, Grants from UCB paid to institution.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (