

Background: Biosimilar therapies have been licensed for treatment of Juvenile Idiopathic Arthritis (JIA) since 2016, and have seen increasing uptake by patients due to the competitive costs compared to their originator products. Several observational studies have been published showing the real-world data of using such biosimilars in JIA patients. However, there is no study consolidating the overall safety and efficacy profile of biosimilars in JIA.
Objectives: To summarize the existing data published to-date on the efficacy and safety profile of different biosimilars of anti-Tumor Necrosis Factor-α (TNFα) in treatment of JIA, compared to their originators.
Methods: A systematic literature search was conducted through the Cochrane Library, Embase, PubMed, Scopus, and Web of Science, to identify studies evaluating the use of anti-TNFα biosimilars in paediatric patients (≤18 years) with JIA compared against either the originators or placebo, published from inception till 30 th August 2024. Quality of included studies was assessed by the Risk Of Bias In Non-randomized Studies of Interventions (ROBINS-I) tool. Efficacy and safety outcomes were summarized descriptively, and meta-analysis was performed where appropriate.
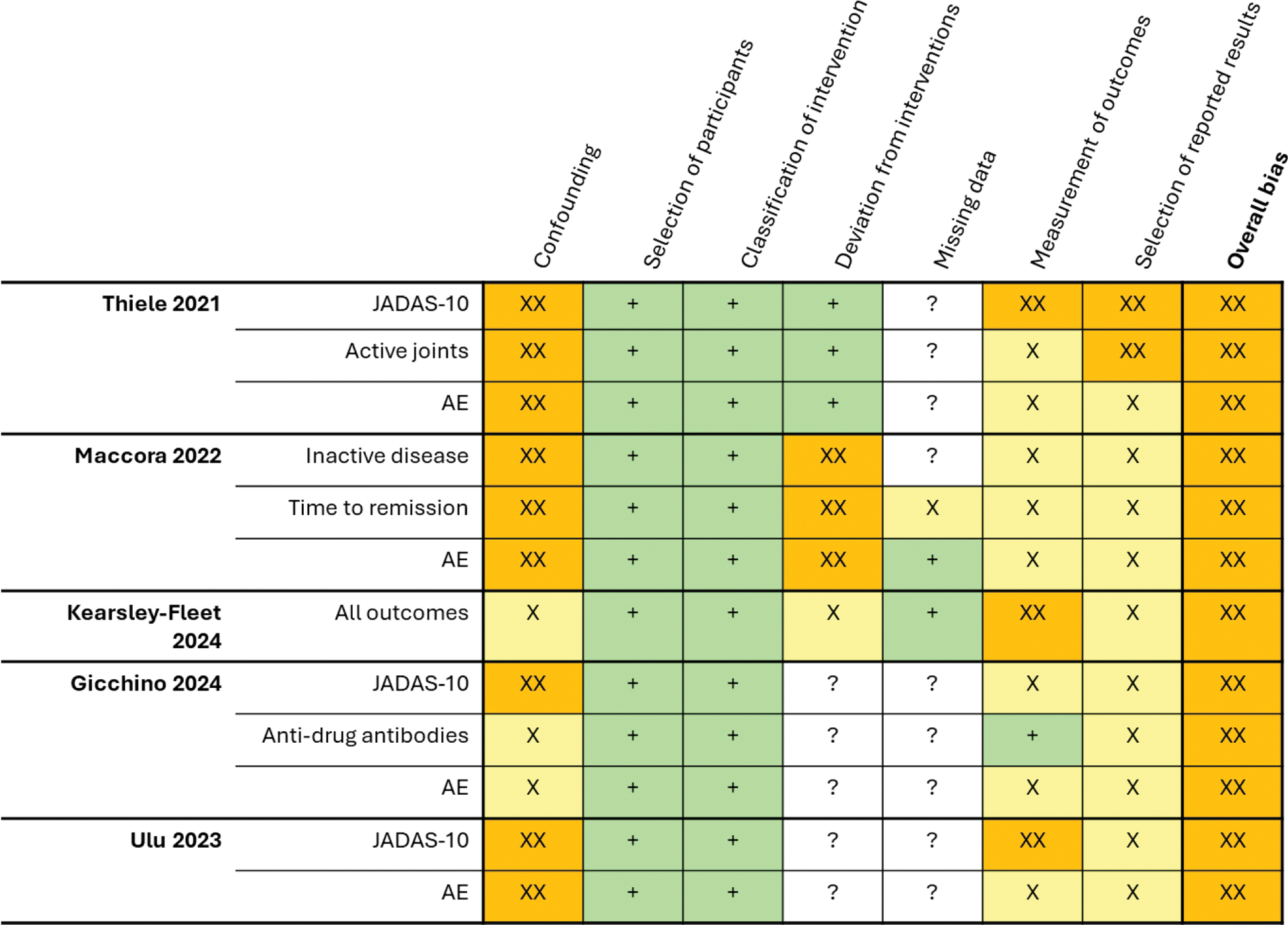
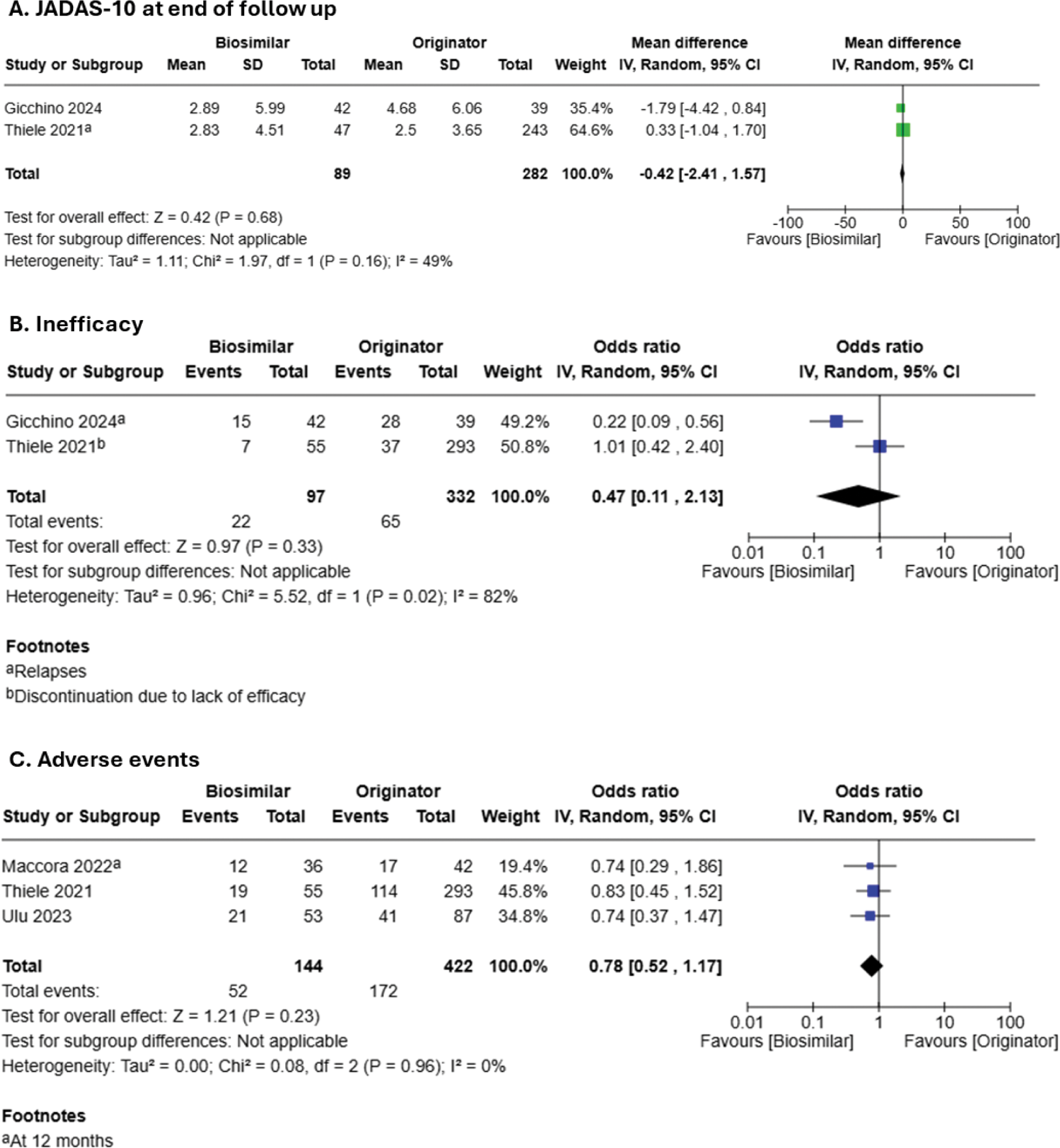
Results: From the 1040 citations assessed, six observational cohort studies were eligible for inclusion, of which one study was excluded from analysis as it did not provide subgroup data on JIA patients. Of the five included studies, two were based on National Registries (Germany and the UK), one on a multicentre cohort and the remaining two on single-centre cohorts. Four studies examined Etanercept, four examined Adalimumab, while one included Infliximab. Quality assessment revealed serious risk of bias in all studies (Figure 1). In terms of efficacy outcomes, all but one study showed no significant difference between biosimilars and originators in head-to-head comparisons of disease activity as measured by active joint counts, JADAS-10, or JADAS-71, etc; time to remission as well as rate of discontinuation due to inefficacy were similar between two groups. Notably, Gucchino and colleagues (2024) demonstrated a better overall profile of Etanercept and Adalimumab biosimilars than their originators, with lower JADAS-10 scores at month 3, 6, 12 and 24. Nonetheless, pooled estimate did not achieve statistical significance, with high level of heterogeneity (Figure 2A). In two studies on switching of originators to biosimilars, disease activities remained stable after switch till end of study follow up. Adverse events (Figure 2C) and drug survival were shown to be similar between biosimilars and originators as well.
Conclusion: Existing real-world data has not shown significant differences in either efficacy or safety between biosimilars and originators of anti-TNFα in treating paediatric patients with JIA. Further studies are needed to generate high quality data on the use of anti-TNFα biosimilars in different subgroups of JIA, with extension to other types of anti-TNFα (e.g. Infliximab, Golimumab).
REFERENCES: [1] Gicchino, M. F., A. N. Olivieri, A. Amodio, G. Capasso, E. Miraglia Del Giudice and A. Di Sessa (2023). “A real-world experience on the efficacy of biosimilar adoption in children with juvenile idiopathic arthritis.” Pediatric Rheumatology 21((Gicchino M.F.; Olivieri A.N.; Amodio A.; Capasso G.; Miraglia Del Giudice E.; Di Sessa A.) Department of Woman, Child and General and Specialized Surgery, University of Campania Luigi Vanvitelli, Napoli, Italy).
[2] Kearsley-Fleet, L., S. Jain, E. Baildam, M. W. Beresford, S. Douglas, H. E. Foster, T. R. Southwood and K. L. Hyrich (2022). “ADALIMUMAB ORIGINATOR VERSUS BIOSIMILAR IN CHILDREN AND YOUNG PEOPLE WITH JIA.” Rheumatology Advances in Practice 6((Kearsley-Fleet L.; Jain S.; Hyrich K.L.) Centre for Epidemiology Versus Arthritis, University of Manchester, Manchester, United Kingdom(Baildam E.; Beresford M.W.) Department of Paediatric Rheumatology, Alder Hey Children’s NHS Foundation Trust, Liverpoo): i57-i58.
[3] Maccora, I., N. Lombardi, G. Crescioli, A. Bettiol, R. Bonaiuti, I. Pagnini, V. Maniscalco, E. Marrani, M. V. Mastrolia, C. Ravaldi, R. Consolini, M. Cattalini, A. Vannacci and G. Simonini (2022). “OBSIDIAN - real-world evidence of originator to biosimilar drug switch in juvenile idiopathic arthritis.” Rheumatology (Oxford) 61(4): 1518-1528.
[4] Thiele, F., A. Klein, A. Hospach, D. Windschall, S. Mrusek, J. M. Ruehlmann and G. Horneff (2021). “Efficacy and Safety of Etanercept Biosimilars Compared With the Originator for Treatment of Juvenile Arthritis: A Prospective Observational Study.” ACR Open Rheumatology 3(11): 779-787.
[5] Ulu, K., M. Çakan, Ş. Çağlayan, R. E. Yiğit, F. Demir, T. Coşkuner, E. Kardeş and B. Sözeri (2023). “Real-life data on efficacy and safety of original Adalimumab and biosimilar Adalimumab (ABP 501) in pediatric rheumatic diseases.” Expert Opinion on Biological Therapy 23(8): 833-839.
Quality assessment of included studies using ROBINS-I tool. AE, adverse events. XXX, critical risk of bias; XX, serious risk of bias; X, moderate risk of bias; +, low risk of bias; ?, no information
Meta-analysis of efficacy and safety outcomes of biosimilars of anti-Tumor Necrosis Factor-α (TNFα) compared with originators in treatment of Juvenile Idiopathic Arthritis (JIA). A, JADAS-10 at the end of follow up; B, Inefficacy, reflected by either relapse or drug discontinuation due to inefficacy; C. Adverse events.
Acknowledgements: We would like to thank Professor Vannacci and his team for kindly providing us with their original research data in the OBSIDIAN study.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (