

Background: Systemic sclerosis (SSc) is a severe multisystem disease with limited treatment options. Case series indicates that anti-CD19 CAR-T cells may be effective in treating SSc; however, their use is limited due to high costs and complex administration [1]. Blinatumomab, a bispecific T-cell engager targeting CD3 and CD19, induces T-cell-mediated depletion of CD19+ B cells. Given its lower cost and simple administration, the potential efficacy of blinatumomab in severe SSc warrants further investigation.
Objectives: To evaluate the safety and effectiveness of blinatumomab in managing severe refractory systemic sclerosis in real-world settings.
Methods: Patients with severe, refractory, Scl70-positive SSc were considered for treatment with blinatumomab following a multidisciplinary review to validate its off-label use. Informed, written consent was obtained from patients to participate in a translational study evaluating the impact of blinatumomab on their immune system. The treatment protocol included premedication with hydrocortisone (100 mg), paracetamol (1 g), and dexchlorpheniramine (5 mg), followed by continuous intravenous infusion of blinatumomab at 9 µg/day for 7 days, then 28 µg/day for another 7 days. Patients were closely monitored in a hospital setting throughout the intravenous treatment phase. The type-I interferon signature was assessed using RT-qPCR with a six-gene panel. Peripheral B cells were magnetically sorted before treatment and three months after treatment, followed by DNA extraction to perform B-cell receptor (BCR) sequencing and analyze the humoral repertoire.
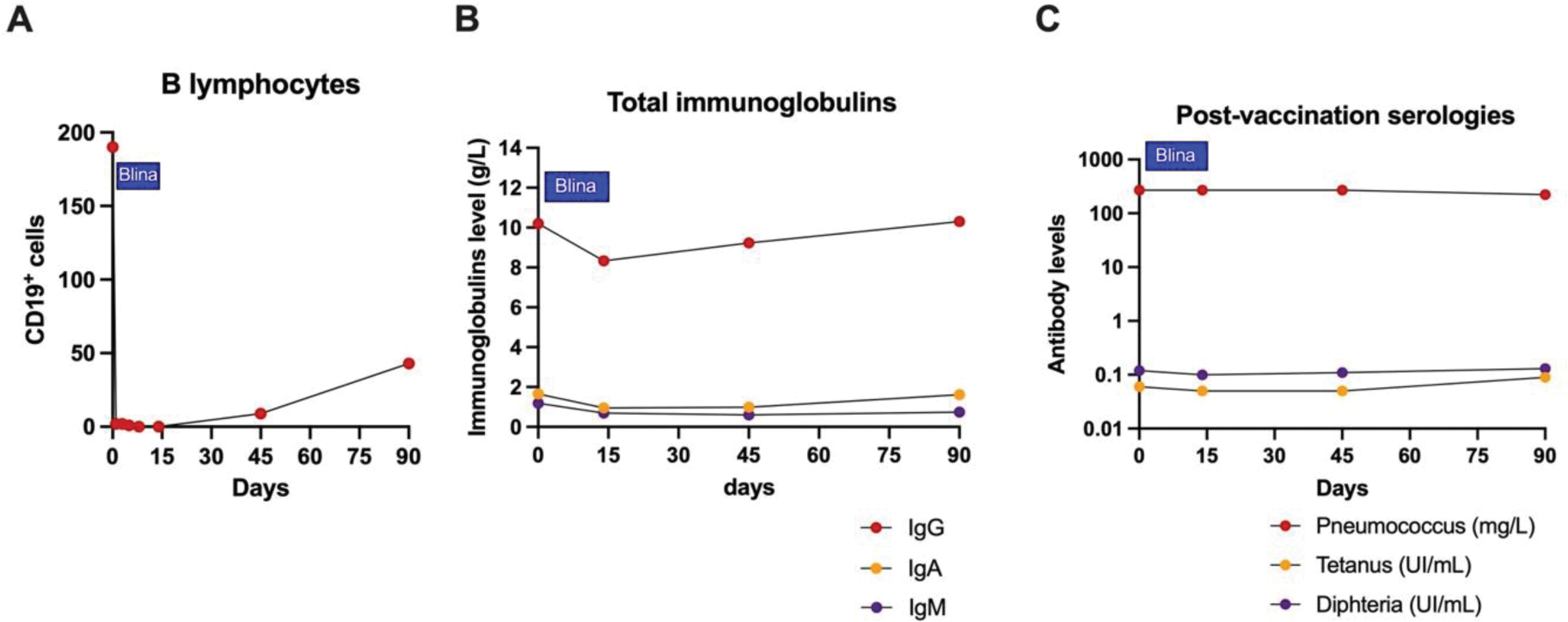
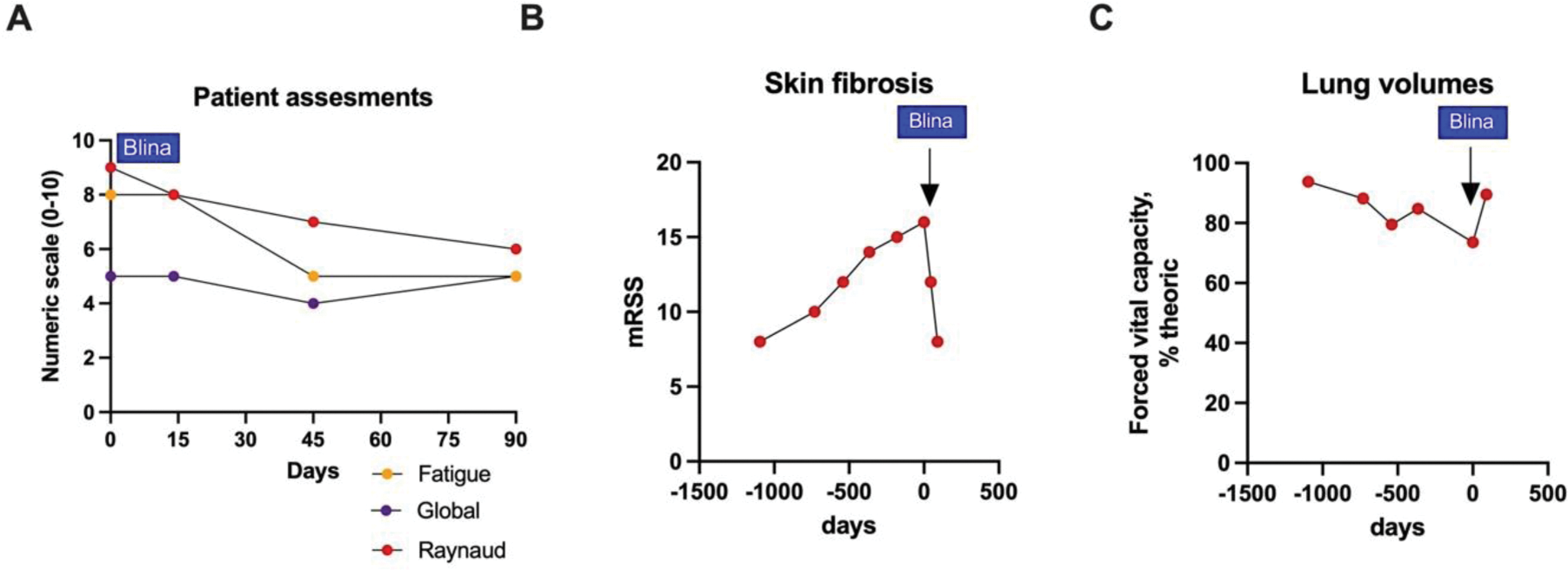
Results: Three patients from three different departments in France were included in the study: three women (P1, P2, and P3) aged 64, 61, and 65 years, respectively. All patients had recent (≤5 years) and progressive Scl70-positive systemic sclerosis (SSc) that was refractory to a median of three immunosuppressive therapies (range: 2–9), including autologous stem cell transplantation for P2. The median modified Rodnan skin score (mRSS) was 16 (range: 5–25). All patients had fibrosing interstitial lung disease; two had cardiac involvement (cardiac fibrosis in P1 and P2; arrhythmia requiring a defibrillator in P1), and none had pulmonary hypertension. At the time of submission, two patients were undergoing blinatumomab treatment (P2, P3), while one (P1) had completed a 3-month post-treatment follow-up. During infusion, P1 experienced a grade I cytokine release syndrome (isolated fever), which was managed with paracetamol without interrupting blinatumomab. No Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS) was observed. Peripheral B-cell depletion was complete by day 3 and persisted until day 14 (Figure 1A). There were no hypogammaglobulinemia, no impact on post-vaccination serologies (Figure 1B-C), and no severe infections. Clinically, P1 reported significant improvements in global disease evaluation, Raynaud’s syndrome, and fatigue (Figure 2A). A notable decrease in mRSS was observed at the 3-month follow-up (8 vs. 16 pretreatment; Figure 2B). While antinuclear and anti-Scl70 antibodies remained unchanged post-treatment, there was a marked reduction in the type I interferon signature. Pulmonary function, as indicated by lung volumes (e.g., forced vital capacity), showed significant improvement after treatment (Figure 2C). B-cell receptor (BCR) sequencing is currently underway.
Conclusion: This real-world study indicates that blinatumomab is safe for use in systemic sclerosis and may lead to clinically significant improvements, including benefits for skin and lung manifestations.
REFERENCES: [1] Scherlinger M et al., The Lancet Rheumatology; 2025, Volume 0, Issue 0; Online First.
Biological impact of blinatumomab treatment (Patient 1). (A) Peripheral CD19+ B cells, (B) total immunoglobulin levels, and (C) post-vaccination serologies.
Clinical impact of blinatumomab treatment (Patient 1). (A) Patient assessment of disease domains, (B) modified Rodnan Skin Score (mRSS), and (C) forced Vital capacity (FVC) expressed in % of the theoric value.
Acknowledgements: The drug was provided and paid by the local university hospital. The pharmaceutical company was not involved in this study.
Disclosure of Interests: Marc Scherlinger Abbvie, Amgen, AstraZeneca, Biogen, BMS, Fresenius, Galapagos, GSK, Innate Pharma, Nordic Pharma, Novartis, Roche, Sandoz, Yannick Dieudonné: None declared, Anna Moltó: None declared, Emmanuel Chatelus: None declared, Aurelien Guffroy: None declared, Thierry Martin: None declared, Jacques-Eric Gottenberg: None declared, Celestine Simand: None declared, Anne El Aatmani: None declared, Eden Sebbag: None declared, Justine Decroocq: None declared, Jerome Avouac: None declared, Jean Sibilia: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (