

Background: Systemic lupus erythematosus (SLE) is an autoimmune disease primarily affecting young women of childbearing age. Autoreactive B cells are central to the pathogenesis of SLE, and variability in B-cell depletion with type I anti-CD20 antibodies in SLE may be responsible for inconsistent clinical responses. Mosunetuzumab is a humanised IgG1 CD20xCD3 bispecific antibody approved for refractory follicular lymphoma that engages and redirects T cells to kill B cells. Due to its mechanism of action, mosunetuzumab treatment may provide an alternative means to manage B-cell populations in blood and tissues of patients with autoimmune diseases, such as SLE.
Objectives: To determine the safety, tolerability, pharmacokinetics (PK) and pharmacodynamics of subcutaneously (SC) administered mosunetuzumab in participants with SLE.
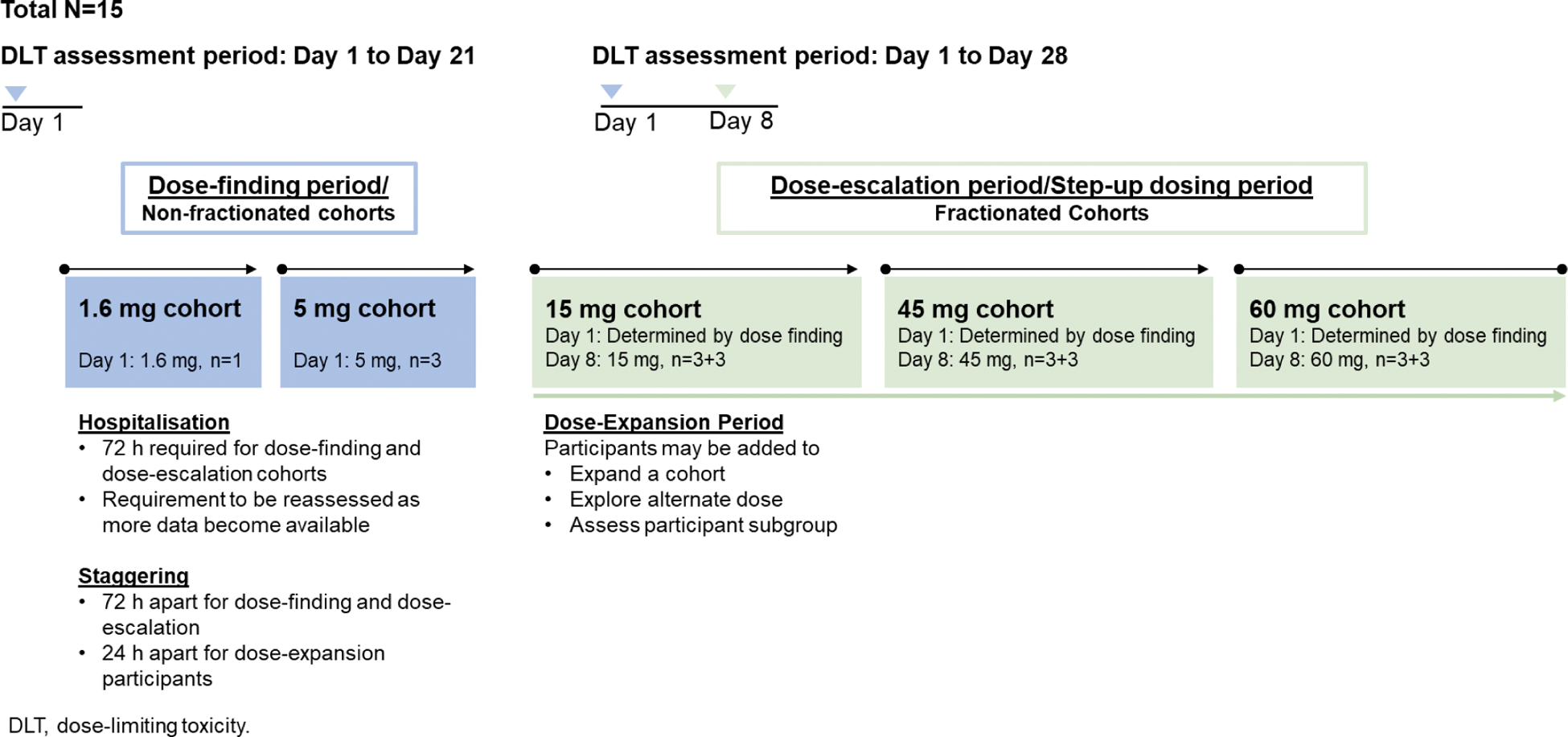
Methods: This Phase Ib, multicentre, open-label, dose-escalation study (NCT05155345) enrolled 15 adult participants diagnosed with active autoantibody positive SLE demonstrated by a Systemic Lupus Erythematosus Disease Activity Index 2000 (SLEDAI-2K) total score ≥4 at screening. The study design included two non-fractionated/dose-finding cohorts and three fractionated/dose-escalation cohorts (Figure 1). To assess severe and unexpected acute drug or injection-related toxicities, a dose-limiting toxicity (DLT) assessment period of 21 days in non-fractionated/dose-finding cohorts and 28 days (21 days after Day 8) in fractionated/dose-escalation cohorts was instituted. All participants were followed for safety for at least 12 months from the last mosunetuzumab dose. Clinical and laboratory assessments, including SLEDAI-2K scores, were also conducted at study visits. Peripheral CD19+ B-cell counts were analysed using high-sensitivity flow cytometry (lower limit of quantification: 0.441 cells/µL). T-cell phenotyping was performed using conventional flow cytometry.
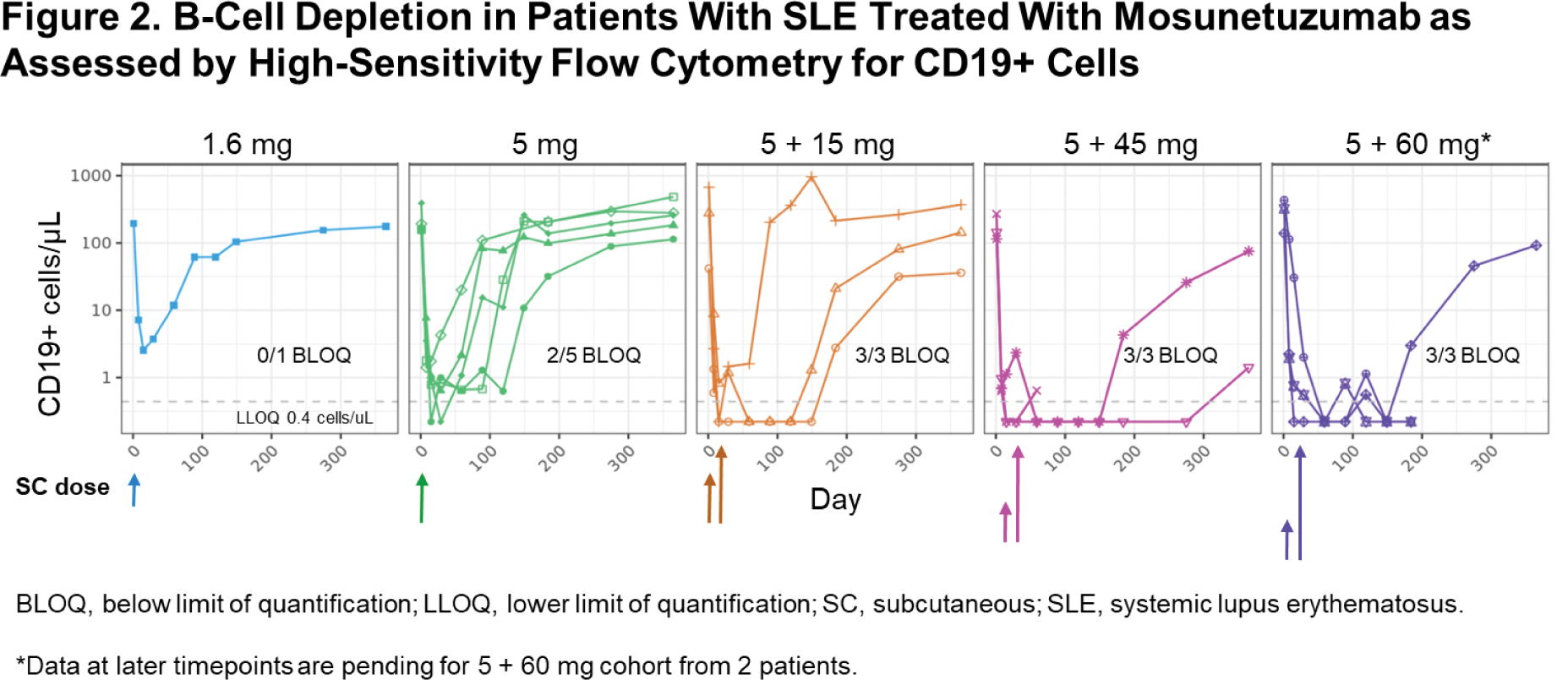
Results: Mosunetuzumab dosing occurred in 15 patients. During the DLT assessment period, no DLTs and no serious adverse events occurred in any cohorts. Four of 15 patients experienced a total of 5 cytokine release syndrome (CRS) events; all were Grade 1-2 and none required tocilizumab administration. During the DLT assessment period, non-serious Grade 1-2 infections occurred in 5 of 15 patients, and no neutropenia or other unexpected high-grade laboratory abnormalities occurred. One patient receiving concomitant acenocoumarol experienced a transient increase in international normalised ratio due to expected cytochrome P450 inhibition by transient cytokine release, requiring acenocoumarol dose adjustment. No high-grade CRS and no immune effector cell-associated neurotoxicity syndrome occurred. One study participant in the 5/45 mg cohort died from interstitial pneumonia 37 days after the end of the DLT assessment period, and the death was deemed unrelated to mosunetuzumab by the investigator and the Sponsor. Four of 6 patients with baseline SLEDAI-2K ≥8 points had a decrease ≥4 points; 3 of 4 patients with positive anti-double-stranded DNA had a decrease in antibody levels. The PK profile of mosunetuzumab SC in patients with SLE appeared consistent with that previously observed for relapsed or refractory Non-Hodgkin lymphoma (NHL) and largely exhibited a dose-dependent increase in exposures. Mosunetuzumab depleted peripheral B cells in patients with SLE (≤0.441 cells/µL) in a dose-dependent manner (Figure 2). Mosunetuzumab-driven T-cell activation revealed that both CD4+ and CD8+ T cells were activated in all tested doses.
Conclusion: Preliminary data from this Phase Ib study indicated that mosunetuzumab exhibits an acceptable safety profile in patients with SLE and that its PK profile in the SLE population is closely aligned with that observed in the relapsed or refractory NHL population. Deep B-cell depletion in the higher-dose cohorts demonstrated the potential for treating B-cell–driven autoimmune diseases. Whilst this study was designed to evaluate the safety and PK profile of mosunetuzumab with a limited dosing regimen, there did appear to be a positive impact on clinical activity. Future clinical studies are required to further evaluate and characterise the efficacy and safety of mosunetuzumab in patients with SLE and other B-cell–driven autoimmune diseases.
REFERENCES: NIL.
Acknowledgements: Funded by F. Hoffmann-La Roche Ltd. Editorial assistance was provided by Nucleus Global, an Inizio company, and funded by F. Hoffmann-La Roche Ltd.
Disclosure of Interests: Vishala Chindalore received research support from F. Hoffman-La Roche Ltd, Elsa Martins employee of F. Hoffmann-La Roche Ltd., William F. Pendergraft III shareholder of F. Hoffmann-La Roche Ltd, employee of Genentech, Inc., X. Rebecca Sheng shareholder of F. Hoffmann-La Roche Ltd., employee of Genentech, Inc., Aaron Schroeder shareholder of F. Hoffmann-La Roche Ltd, employee of Genentech, Inc., Liudmila Gearhart employee of F. Hoffmann-La Roche Ltd, Hardik Mody shareholder of F. Hoffmann-La Roche Ltd., employee of Genentech, Inc., Huiyan (Ashley) Mao shareholder of F. Hoffmann-La Roche Ltd., employee of Hoffmann-La Roche Ltd., Jay Garg shareholder of F. Hoffmann-La Roche Ltd, employee of Genentech, Inc., Iwona Dankiewicz Fares: None declared, Svetlana Agachi received research support from F. Hoffman-La Roche Ltd.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (