

Background: Smoking has been associated with reduced efficacy and higher rates of therapy discontinuation for methotrexate (MTX) and anti-TNF agents in patients with rheumatoid arthritis (RA) and spondyloarthritis (SpA) [1, 2]. However, the potential effects of the interaction between smoking (which can reduce folate levels [3]) and MTX on the safety of anti-TNF therapies have not been extensively studied. We found no prior studies investigating the potential interaction between MTX and smoking on the incidence of serious adverse events and infections in rheumatic patients undergoing treatment with biologic agents.
Objectives: To evaluate the effect of the combination of smoking and methotrexate on the overall safety of anti-TNF therapy in the treatment of RA and SpA, as assessed by the incidence of serious adverse events (SAEs).
Methods: BiobadaBrasil is a multicentric registry-based cohort study of Brazilian patients with rheumatic diseases starting their first biologic DMARD or a Janus kinase inhibitor. The present analysis includes only biologic-naïve RA and SpA patients (recruited from Jan 2009 to Oct 2019) on their first course of an anti-TNF agent (latest date of follow-up, Nov 19, 2019). A treatment course is defined as a period during which the medication scheme does not change, except for dose adjustments. The primary outcome was the incidence of the first SAE. The incidence of the first adverse event of any kind, total infections, and serious infectious served as secondary outcomes. The incidence of treatment interruption due to adverse events also served as secondary outcome. The primary analysis plan aimed to evaluate the impact of smoking on the risk of adverse events, with patients stratified based on their methotrexate use. Kaplan-Meier survival curves, as well as univariate and multivariate Cox proportional hazards models to estimate crude (cHR) and adjusted hazard ratios (aHR), respectively, were used for statistical analyses. Multivariate analyses were adjusted for a broad range of variables representing demographic and clinical characteristics.
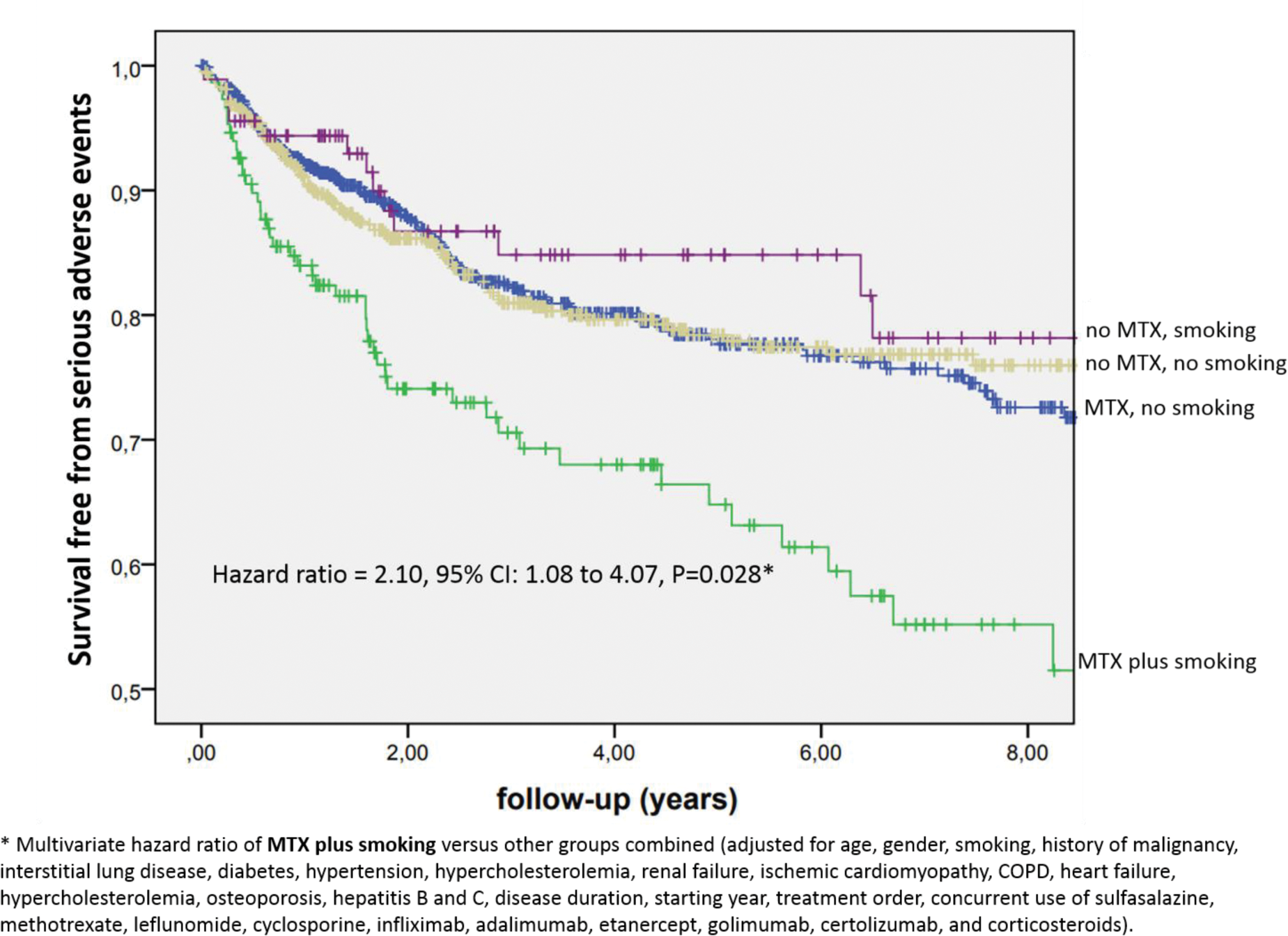
Results: A total of 1,689 patients were enrolled, accounting for 5,780 patient-years (PY) of follow-up. The prevalence of smoking was 14.2%, and methotrexate (MTX) use was reported in 57.0% of patients. The most frequent diagnoses were rheumatoid arthritis (59.9%), ankylosing spondylitis (26.5%), and psoriatic arthritis (10.4%). The anti-TNF agents prescribed were adalimumab (35.3%), infliximab (29.7%) etanercept (26.2%), certolizumab (5.0%) and golimumab (3.8%). The overall incidence of SAEs was 5.31/100 PY. In the entire sample of patients, the incidence of SAEs was associated with smoking in univariate analysis (cHR= 1.50, 95% CI: 1.13–1.98, P=0.005) and multivariate analysis (aHR= 1.48, 1.10–1.99. p=0.009). After stratification according to treatment with MTX, smoking was related to higher risk of SAEs in patients receiving MTX (cHR = 1.98, 1.42–2.76, P<0.001; aHR = 1.77, 1.24–2.53, P=0.002), but not in non-MTX users (cHR = 0.83, 0.48–1.45, P=0.510; aHR =0.85, 0.47–1.53, P=0.584). The results were similar in RA and SpA patients. The results on the risk of smoking were divergent between MTX users and non-users for total adverse events (cHR = 1.42, 95% CI: 1.15–1.77, P = 0.001 vs. cHR = 1.00, 95% CI: 0.73–1.36, P = 0.994), total infections (cHR = 1.52, 95% CI: 1.15–2.01, P = 0.003 vs. cHR = 1.05, 95% CI: 0.68–1.62, P = 0.818), and serious infections (cHR = 2.04, 95% CI: 1.28–3.25, P = 0.003 vs. cHR = 0.93, 95% CI: 0.42–2.06, P = 0.862). The results on the risk of smoking for the outcome of treatment interruption due to adverse events did not differ significantly between MTX users (cHR = 1.38, 95% CI: 0.93–2.03, P = 0.109) and non-users (cHR = 0.96, 95% CI: 0.54–1.71, P = 0.885).The observed association of smoking and SAEs was retested (in the entire sample) including an interaction term of MTX-smoking (besides each of these variables included individually in the model). In this analysis, the interaction term MTX-smoking kept statistically significant association with SAEs (cHR=2.39, 1.25–4.57, p=0.008; aHR=2.01, 1.08–4.07, P=0.028; see Figure 1). However, smoking alone was not associated with SAEs when adjusted for the combination MTX-smoking in this analysis (cHR=0.83, 0.47–1.44, P=0.500; aHR=0.89, 0.50–1.57, P=0.686).
Conclusion: In this study, the use of methotrexate combined with smoking was associated with higher risk of serious adverse events in patients with RA and SpA treated with anti-TNF agents. We did not observe an independent increase in the risk of serious adverse events with smoking in patients not taking MTX. These results require confirmation through further studies, but it is possible that the deleterious effects of smoking in these patients are mediated, at least in part, by its interaction with methotrexate
REFERENCES: [1] Nayebirad S et al. Mod Rheumatol 2023;34(1):68-78.
[2] Ciurea A et al. Ann Rheum Dis 2016;75:532-9.
[3] Salazar J et al. Br J Clin Pharmacol 2024;90:1301-1311.
Acknowledgements: Brazilian Society of Rheumatology and Patricia Cabral (monitor).
Disclosure of Interests: Markus Bredemeier: None declared, Alice de Moraes Baier: None declared, Carolina Vieira Tavares: None declared, Angela Duarte: None declared, Marcelo Pinheiro Abbvie, Janssen, UCB, Abbvie, Janssen, UCB, Barbara Stadler Kahlow: None declared, Monica Valeria Siqueira De Vechi: None declared, Roberto Ranza: None declared, Jose Roberto Miranda: None declared, Valéria Valim: None declared, Glaucio Castro Abbvie, Lilly, Janssen, UCB, Manoel Bertolo: None declared, Maria de Fátima Sauma: None declared, Vander Fernandes: None declared, Ana Cristina Medeiros-Ribeiro: None declared, Reginaldo Botelho: None declared, Claiton Brenol: None declared, Hellen Mary da Silveira de Carvalho: None declared, Samia Studart: None declared, Geraldo da Rocha Castelar Pinheiro: None declared, Laurindo Rocha Jr: None declared, Hugo de Leon de Lima: None declared, Ivanio Pereira: None declared, Morgana Ohira Gazzeta: None declared, Adriana Maria Kakehasi Abbvie, Lilly, Janssen, Organon, Pfizer, UCB, Abbvie, Janssen, Organon, Pfizer, Abbvie, Novartis, Pfizer, UCB, Paulo Louzada Jr: None declared, Andre Luiz Shinji Hayata: None declared, Cristiano Lupo: None declared, Ines Guimaraes Silveira: None declared, Sergio Kowalski: None declared, David Titton: None declared, Aline Ranzolin: None declared, Ricardo Machado Xavier Abbvie, UCB, Janssen, Organon, Abbvie, UCB, Ieda Maria Laurindo Abbvie, Janssen, Abbvie, Bristol, Janssen, Novartis, Pfizer, UCB, Abbvie, Boering, Bristol, Janssen, Lilly, Pfizer, UCB, Jose Eduardo Martinez: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (