

Background: Systemic lupus erythematosus (SLE) is a systemic autoimmune disease affecting 43.7 (15.87 to 108.92) per 100,000 persons worldwide and might be potentially life-threatening. Anifrolumab is a monoclonal antibody that blocks interferon-alpha receptors and has demonstrated efficacy in clinical trials in SLE patients [1]. This medication demonstrated significant efficacy in clinical trials. The RELES registry is an inception cohort including SLE patients diagnosed from 2009 [2]. The registry includes 969 SLE patients treated by specialized doctors of the Internal Medicine department in 50 centers in Spain. Here, we present the preliminary data of the patients treated with anifrolumab in the registry.
Objectives: Primary Objective: To describe the clinical effectiveness of anifrolumab defined by the proportion of patients that achieve LLDAS or remission, measured by DORIS-21. Secondary Objectives:
- To describe the clinical effectiveness of anifrolumab over time in routine clinical practice defined by disease activity, measured by SLEDAI and PGA.
- To describe clinical SLE manifestations considering flares (that are defined as new or persistent clinical signs in the SLEDAI score and the absence of LLDAS) and organ damage (measured as SLICC/ACR Damage Index).
Methods: We performed a retrospective multicenter inception cohort analysis of the patients included in the RELES registry treated with anifrolumab. At least one visit was completed once yearly from the registry inclusion in every patient. We selected a visit 6 months before the treatment onset and the visit that led to the onset of anifrolumab treatment. After that, we registered a visit 6 months after the anifrolumab onset (or the nearest if unavailable). Clinical involvement, activity scores (SLEDAI and PGA), SLICC score and presence of flares were recorded every visit. We used the t-student test for related variables to compare SLEDAI, PGA, and the SLICC score before and after the anifrolumab treatment. The McNemar test was used to compare the DORIS remission and the LLDAS before and after anifrolumab treatment. The Ethics committee approved the protocol (IB 1246/09 PI). The registry received funding from AstraZeneca.
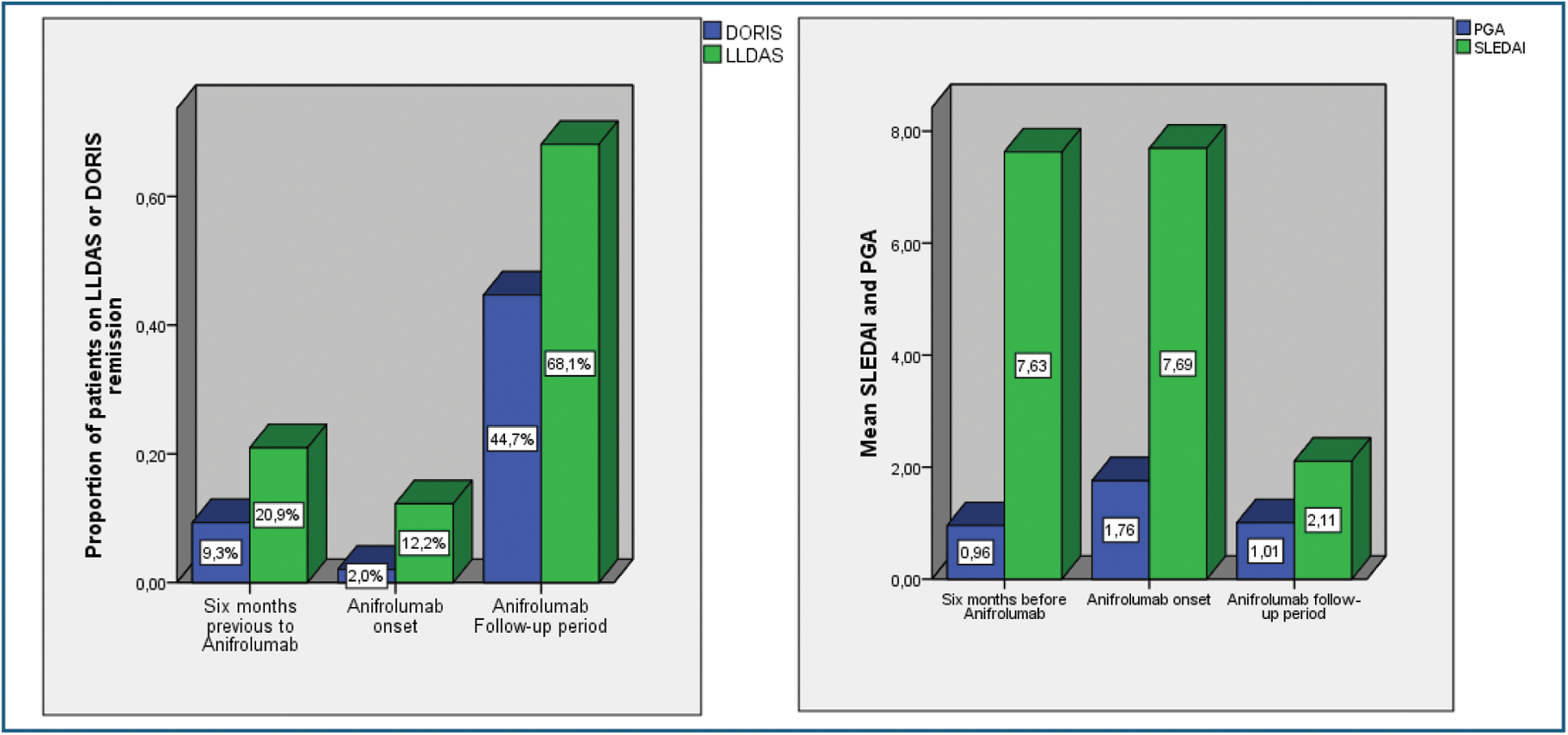
Results: Of the 969 SLE patients included in the RELES registry from 2009, 49 were treated with anifrolumab and included for analysis. The mean age at anifrolumab initiation was 44 years, and 87.75% were women (43/49). The mean time from diagnosis to the anifrolumab treatment was 9.9 years (median 7.74 years). The proportion of patients in DORIS remission at the onset of anifrolumab treatment was 2% (1/49) and significantly increased to 44.7% (21/47). Similarly, the LLDAS rose from 12.2% (6/49) to 68.1% (32/47) during the anifrolumab follow-up time (Figure 1). In the first 6 months before the anifrolumab onset, the mean (SD) SLEDAI was 7.63 (4.58), and the mean (SD) PGA was 0.96 (1.1). At the start of the anifrolumab treatment, the mean (SD) SLEDAI was 7.69(3.98), and the PGA was 1.76(0.60). After the follow-up period under anifrolumab treatment, the SLEDAI decreased to 2.11(2.93), p<0.001), and the PGA was reduced to 1.01(0.95), p<0.001. (Figure 1). Most patients had cutaneous involvement in 95.9% (47/49) with subacute cutaneous lupus, discoid lupus, cutaneous vasculitis or photosensitivity. A total of 73.5% presented joint involvement through the visits and 57% had leucopenia. An 87.8% (43/49) of patients were flared when they started the anifrolumab treatment. At the end of the follow-up time with anifrolumab, 31.9% (15/47) of patients had presented at least one flare during the follow-up. Among these 15 patients, two patients withdrew anifrolumab due to inefficacy. The other 13 patients continued anifrolumab treatment. Chronic damage measured by the mean (standard deviation [SD]) SLICC score was 0.51(0.84) at the anifrolumab onset. This score did not increase during the anifrolumab treatment follow-up with a mean (SD) SLICC score of 0.55(0.88). Only two patients increased their SLICC score by one point under anifrolumab treatment. The mean anifrolumab treatment follow-up time was 212.6 ± 119 days.
Conclusion: Six-month follow-up data of anifrolumab treatment in systemic lupus erythematosus in a real-world setting show a good response to maintain remission and a low disease activity state. The drug also demonstrated decreased disease activity, measured by SLEDAI and PGA. Anifrolumab showed a good response in the clinical trial settings. Here, we also provide a positive clinical evolution with real-world data. Data including further follow-up would be needed to describe the use of anifrolumab in LES in Spain.
Proportion of patients on LLDAS and DORIS remission (A). Mean SLEDAI and PGA (B)
REFERENCES: [1] Morand EF, Furie R, Tanaka Y, Bruce IN, Askanase AD, Richez C, Bae SC, Brohawn PZ, Pineda L, Berglind A, Tummala R; TULIP-2 Trial Investigators. Trial of Anifrolumab in Active Systemic Lupus Erythematosus. N Engl J Med. 2020 Jan 16;382(3):211-221. doi: 10.1056/NEJMoa1912196. Epub 2019 Dec 18. PMID: 31851795 .
[2] Canora J, García M, Mitjavila F, Espinosa G, Suárez S, González-León R, Sopeña B, Boldova R, Castro A, Ruiz-Irastorza G; en representación de los investigadores del Registro RELES-Grupo de Enfermedades Autoinmunes Sistémicas (GEAS). Clinical characteristics during diagnosis of a prospective cohort of patients with systemic lupus erythematosus treated in Spanish Departments of Internal Medicine: The RELES study. Rev Clin Esp (Barc). 2017 Jan-Feb;217(1):7-14. English, Spanish. doi: 10.1016/j.rce.2016.09.006. Epub 2016 Oct 25. PMID: 27793331.
Acknowledgements: Marta Galvez Fernandez.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (