

Background: Gout is associated with increased cardiovascular (CV) risk. This is in part due to traditional CV risk factors, however, gout itself is also an independent risk factor. Both (changes in) serum urate concentrations and systemic and vascular inflammation in between and during gout flares putatively contribute to the excess CV risk associated with gout. Mainstay of long-term gout treatment consists of urate-lowering therapy (ULT), with allopurinol being the most commonly used. The effect of allopurinol on CV risk in gout patients has been the subject of debate. As some studies suggest a general protective effect of ULT on CVD [1], where others do not find any CV risk reduction with allopurinol in patients with ischaemic heart disease but no gout [2]. The effect of allopurinol might however be disease- and also time- and treatment stage dependent. In addition to ULT use, colchicine is used to treat gout flares and is also recommended as flare prophylaxis during the first months of treatment with ULT [3]. The protective effect of colchicine on CV risk is subject of debate as well, and this might also differ between ULT treatment stages.
Objectives: The aim of this observational prescription database study was therefore to investigate the associations of allopurinol use with CV risk during different stages of treatment, including initiation, chronic use and discontinuation of allopurinol, with a focus on the potential modifying effect of allopurinol dose and concomitant colchicine use.
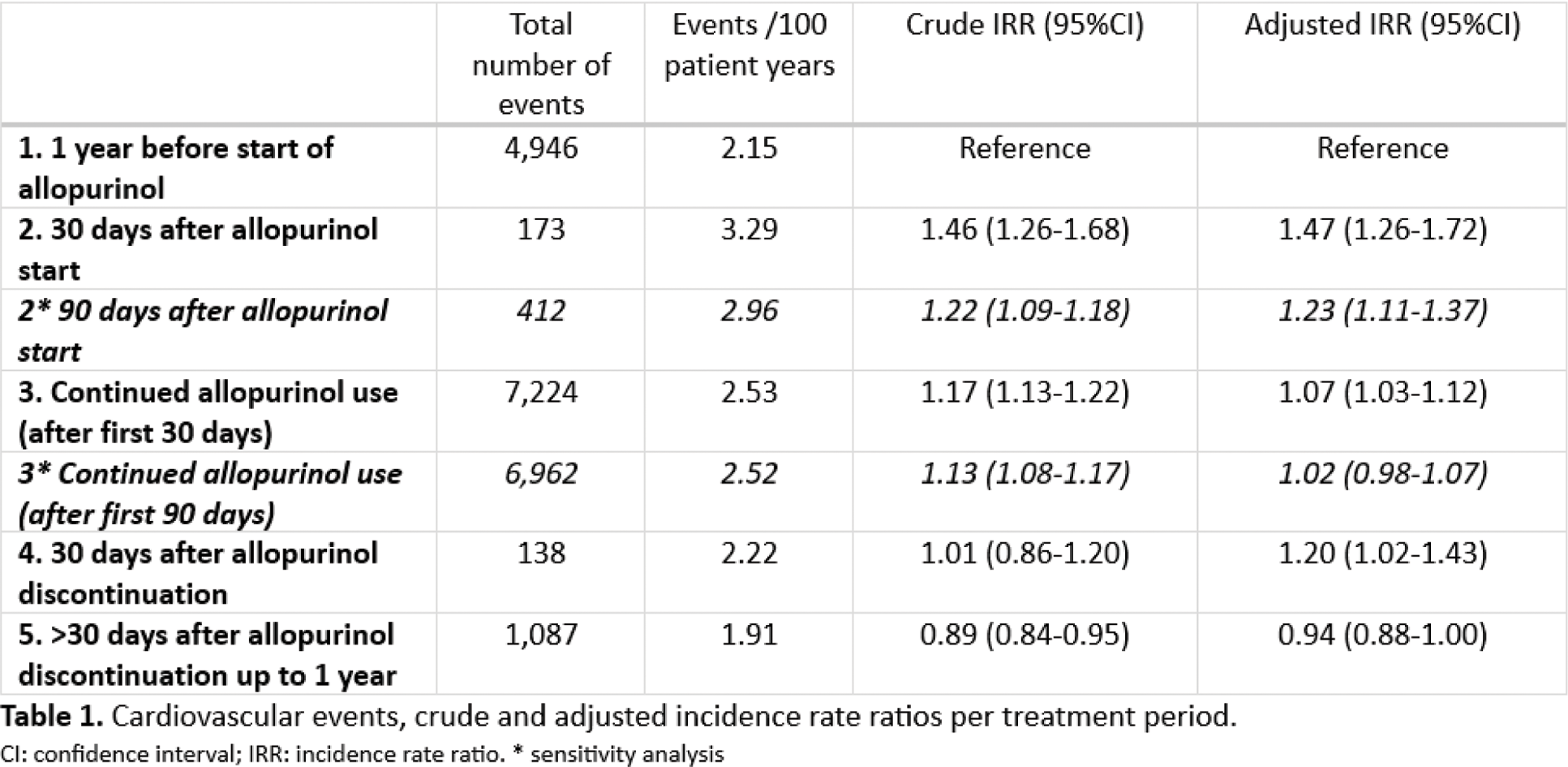
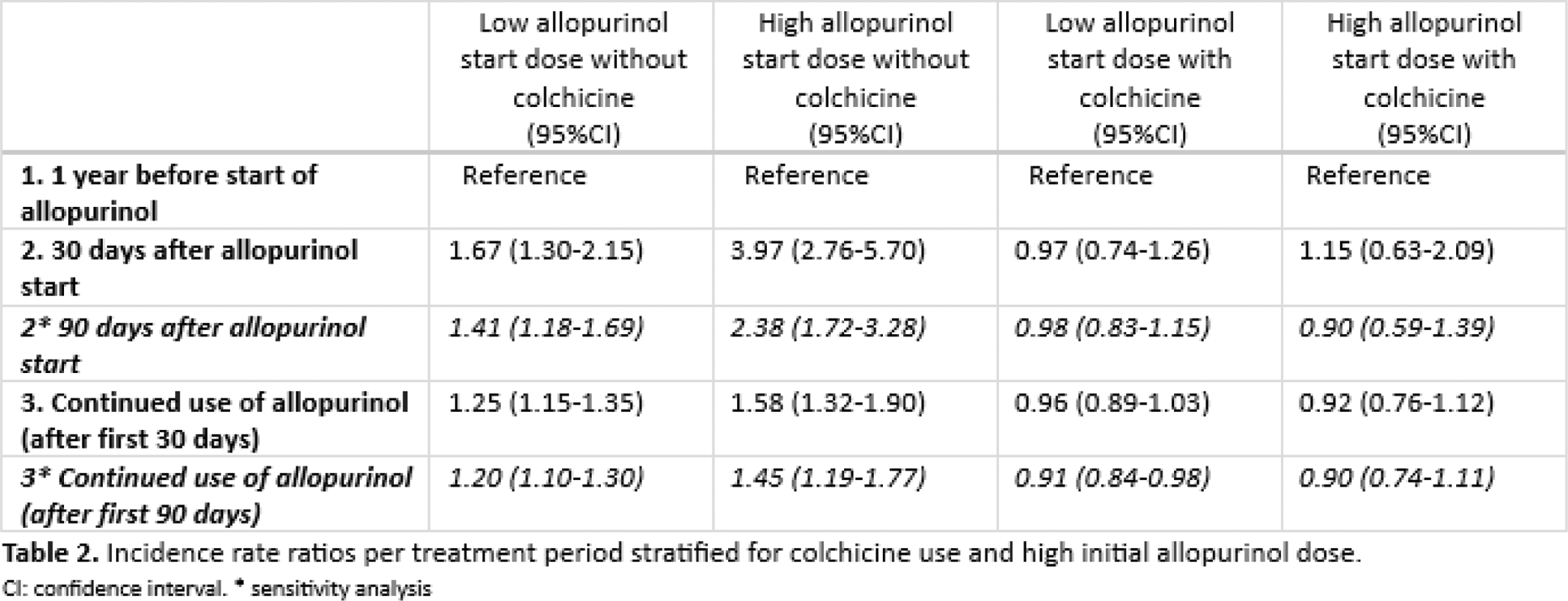
Methods: A pharmaco-epidemiologic cohort study, including adult patients prescribed allopurinol between October 2015 and December 2022. To identify patients with gout, dispensed prescriptions had to be made by either a rheumatologist, general practitioner, internal medicine specialist, elderly care physician or geriatrician. A CV event was defined as a new dispensed prescription for a platelet aggregation inhibitor (clopidogrel, prasugrel, ticagrelor or dipyridamole). Incidence densities (CV events/100 patient years) were compared using incidence rate ratios (IRRs). Five time periods were predetermined: period 1, 1 year before first allopurinol initiation; period 2, the first 30 days after initiating allopurinol (first 90 days as sensitivity analysis); period 3, during continued allopurinol use; period 4, the first 30 days after allopurinol discontinuation; and period 5, prolonged discontinuation of allopurinol up to 1 year. A first prescription of allopurinol was identified when a patient picked up other prescriptions but no allopurinol before initiating allopurinol. Allopurinol discontinuation was defined as patients who stopped picking up prescriptions for allopurinol, at least one year before December 31 st 2022 (end of cohort period) or a time gap between prescription stop date and start >90days. Multilevel Poisson regression analysis was used to estimate the incidence rate ratios of CV events for the five time periods, both crude and adjusted for potential confounders (age, sex, diabetes mellitus, hypertension and hypercholesterolemia), with period 1 (for allopurinol start analyses) or 4 (for allopurinol stop analyses) as reference. Effect modification by initial allopurinol dose — low (<300 mg) or high (≥300 mg) — and concomitant colchicine use (at least 80% use during period 2) was assessed.
Results: Included were 84,582 patients (77% male, mean age 66 years (sd 13.4)), with 122,659 treatment episodes and 13,568 CV events. CV risk for periods 2, 3, 4 and 5 (reference period 1) were: 1.47 (95%CI 1.26-1.72), 1.07 (95%CI 1.03-1.12), 1.20 (95%CI 1.02-1.43), and 0.94 (95%CI 0.88-1.00) respectively (Table 1). The increased CV risk after starting allopurinol was dose-dependent (IRR: low dose 1.67; 95%CI 1.30-2.15; high dose 3.97; 95%CI 2.76-5.70) (table 2). This increase was fully countered by concomitant use of colchicine (IRR low dose allopurinol with colchicine 0.97; 95%CI 0.74-1.26, high dose allopurinol with colchicine 1.15; 95%CI 0.63-2.09) (table 2).
Conclusion: After allopurinol start – especially the first 30 to 90 days - there is a dose-dependent increase in the risk of cardiovascular risk, which is fully countered by concomitant colchicine use. Discontinuation of allopurinol does not lead to a relevant increase in cardiovascular event risk. Concomitant use of colchicine or slower dose escalation of allopurinol seems prudent to decrease the risk of CV events in gout patients starting allopurinol. These data support the current recommendation to use concomitant colchicine when starting allopurinol, or alternatively, use a low starting dose of allopurinol.
REFERENCES: [1] Bredemeier M, Lopes LM, Eisenreich MA, al. e. Xanthine oxidase inhibitors for prevention of cardiovascular events: a systematic review and meta-analysis of randomized controlled trials. BMC Cardiovasc Disord. 2018;18(1):24.
[2] Mackenzie IS, Hawkey CJ, Ford I, al. e. Allopurinol versus usual care in UK patients with ischaemic heart disease (ALL-HEART): a multicentre, prospective, randomised, open-label, blinded-endpoint trial. Lancet. 2022;400(10359):1195-205.
[3] Richette P, Doherty M, Pascual E, al. e. 2016 updated EULAR evidence-based recommendations for the management of gout. Ann Rheum Dis. 2017;76(1):29-42.
Acknowledgements: NIL.
Disclosure of Interests: Iris Rose Peeters: None declared, Maike H.M. Wientjes: None declared, Isla S. Mackenzie Outside the submitted work, Outside the submitted work, Outside the submitted work, Merel Opdam: None declared, Marcel Flendrie: None declared, Calin Popa: None declared, Kayleigh M. van de Wiel: None declared, Dina Vojinovic: None declared, Leo Joosten: None declared, Bas Schouwenberg: None declared, Martijn Gerritsen: None declared, A.A. den Broeder: None declared, Noortje van Herwaarden: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (