

Background: Anti-HMGCR antibodies are a biomarker of immune-mediated necrotising myopathy (IMNM), a subtype of inflammatory myopathies (IM) characterised by weakness and myofibre necrosis whose mechanism is currently unknown [1]. It has recently been shown that anti-HMGCR antibodies are internalized into the myofibres of IMNM patients [2]. Several observations indicate that this may result in disrupted HMGCR function and a subsequent myopathic effect: (i) anti-HMGCR antibodies target the HMGCR enzymatic active site [3]; (ii) both the statin inhibition of HMGCR (4) and mutations impairing its activity [4] lead to myofibre necrosis resembling IMNM [1]; (iii) lipid droplets accumulation (a marker of fatty acid beta-oxidation defect) resembling statin myopathy has been observed in myofibres of anti-HMGCR patients [2]. However, no direct evidence of this hypothesis has been reported to date [2].
Objectives: The aim of this study was to investigate whether anti-HMGCR antibodies interfere with HMGCR activity and have a myopathic effect.
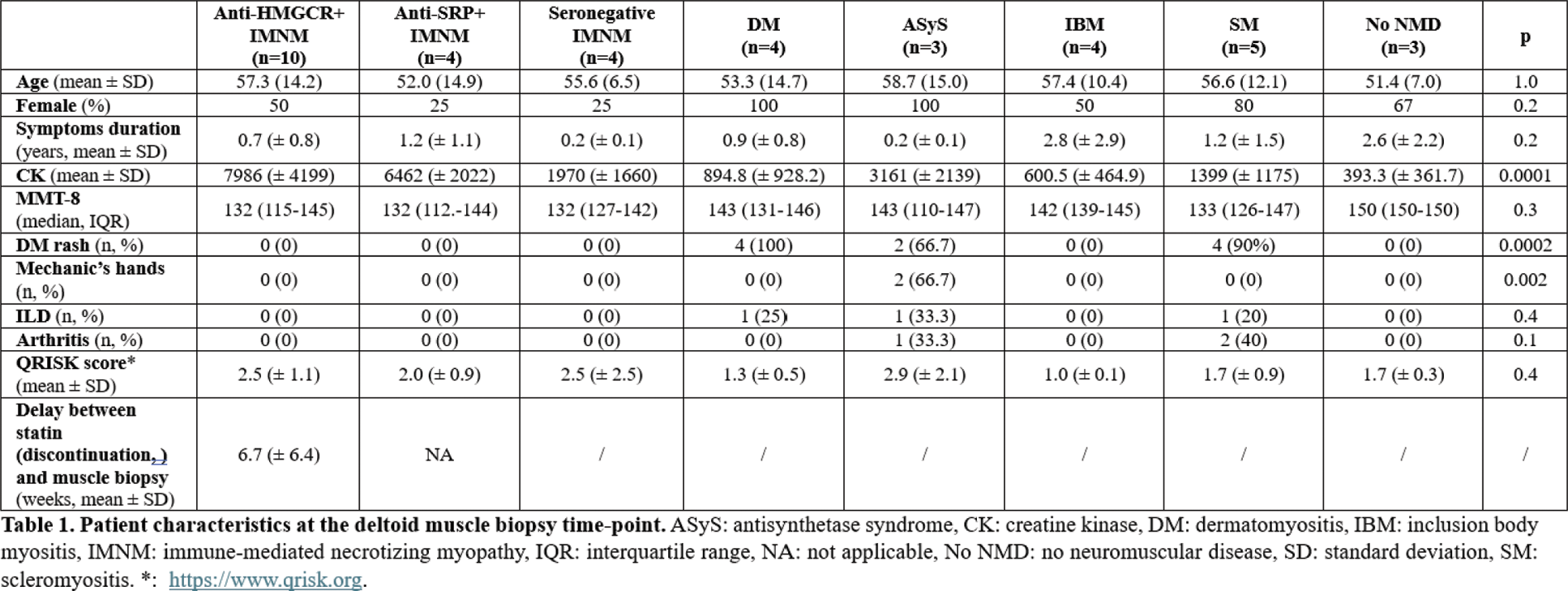
Methods: To obtain polyclonal anti-HMGCR autoantibodies, the 6 peptides identified as the epitopes of anti-HMGCR antibodies [3] were synthesised and used to immunise a white New Zealand rabbit. Antibodies from a non-immunised animal served as control. Additionally, plasma of anti-HMGCR (n=5) and anti-SRP (n=2) IMNM patients were obtained from plasmapheresis eluates performed for therapeutic purposes. Rabbit and human anti-HMGCR antibodies were purified by affinity chromatography. Inhibition of HMGCR was measured in vitro by spectrophotometry in the presence of different concentrations of autoantibodies. Pravastatin was used as a negative control. Human autoantibodies were electroporated into human myotubes, and hematoxylin/eosin (H&E) and oil red-O stainings were performed. Thirty-four patients with IM according to the EULAR/ACR 2017 criteria, not taking immunomodulators or statins, were prospectively included (anti-HMGCR: n=10; other IM: n=24) as well as 3 patients without neuromuscular diseases (no NMD). Histological sections of deltoid muscle taken at the time of diagnosis were immunostained with an anti-human IgG antibody. Oil red-O staining was used to study the accumulation of lipid droplets, quantified according to an extension score (0-4) by two myopathologists blinded to the diagnosis. The metabolites of the cholesterol synthesis pathway directly upstream (HMG-CoA) and downstream (mevalonate) of HMGCR were determined by mass spectrometry in serum and muscle of patients.
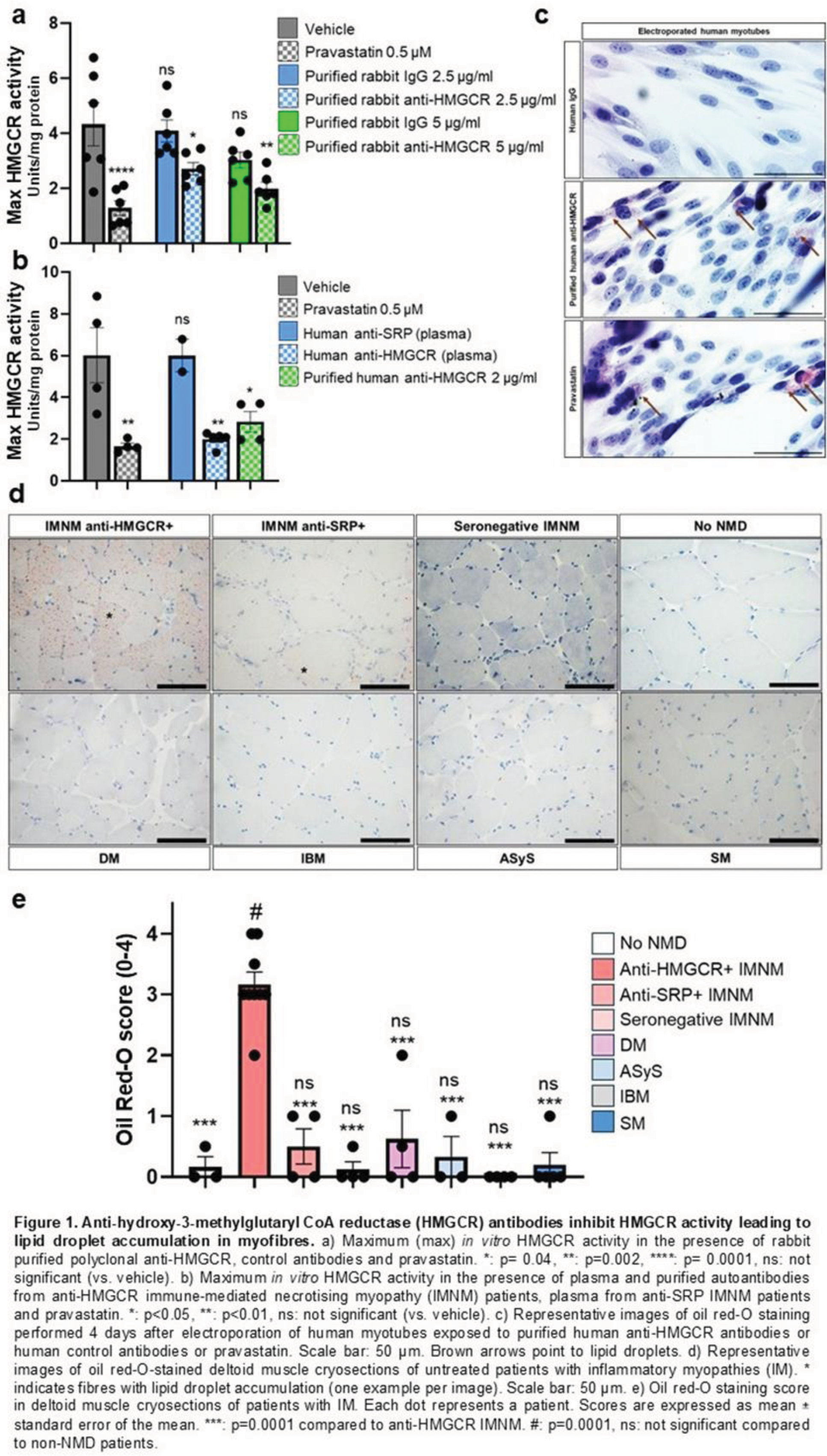
Results: To test whether anti-HMGCR antibodies inhibit the function of their target, HMGCR enzymatic activity was assessed in vitro in the presence of anti-HMGCR, control antibodies and pravastatin. A dose-dependent inhibition of HMGCR activity was observed in the presence of anti-HMGCR (Figure 1a). Rabbit anti-HMGCR 5 µg/ml and human anti-HMGCR 2 µg/ml had similar effects to that of pravastatin 0.5 μM (Figure 1a, 1b). IgG from the non-immunised rabbit and plasma from anti-SRP patients did not affect HMGCR activity (Figure 1a, 1b). To test whether anti-HMGCR internalisation in myofibres exerts a myopathic effect, human myotubes were electroporated with human purified anti-HMGCR, control IgG or pravastatin. Antibody presence in the cytoplasm 4 days after electroporation was confirmed. H&E staining showed necrotic human myotubes after electroporation with purified anti-HMGCR and pravastatin, but not with control IgG. Oil red-O staining revealed a lipid droplet accumulation in human myotubes electroporated with purified anti-HMGCR and pravastatin, but not with control IgG (Figure 1c). This finding was also observed in patients whose characteristics are presented in Table 1. Lipid droplet accumulation score was ten-fold higher in anti-HMGCR IMNM patients compared to other IM and no NMD patients (3.2 ± 0.6 vs. 0.3 ± 0.5; vs. 0.3 ± 0.6, respectively, p=0.0001) and a score ≥ 2 was a hallmark of anti-HMGCR IMNM (Figure 1d, 1e). The increase of HMG-CoA and the decrease of mevalonate in the muscle and serum of anti-HMGCR patients indicated that HMGCR activity was blocked in the muscle of anti-HMGCR patients and not in the other IM nor in no NMD patients.
Conclusion: Together, these data demonstrate that anti-HMGCR antibodies inhibit HMGCR activity leading to accumulation of myopathic lipid droplets in myofibres. These findings could have potential implications for both the diagnosis and treatment of IMNM.
REFERENCES: [1] Allenbach Y, et al. Immune-Mediated Necrotizing Myopathies Working Group. 224th ENMC International Workshop: Clinico-sero-pathological classification of immune-mediated necrotizing myopathies Zandvoort, The Netherlands, 14-16 October 2016. Neuromuscul Disord NMD. 2018 Jan;28(1):87–99.
[2] Pinal-Fernandez I, et al. Pathological autoantibody internalisation in myositis. Ann Rheum Dis. 2024 Jun 20;ard-2024-225773.
[3] Musset L, et al. Analysis of autoantibodies to 3-hydroxy-3-methylglutaryl-coenzyme A reductase using different technologies. J Immunol Res. 2014;2014:405956.
[4] Yogev Y, et al. Limb girdle muscular disease caused by HMGCR mutation and statin myopathy treatable with mevalonolactone. Proc Natl Acad Sci. 2023 Feb 14;120(7):e2217831120.
[5] Liepinsh E, et al. Hydroxymethylglutaryl-CoA reductase activity is essential for mitochondrial β-oxidation of fatty acids to prevent lethal accumulation of long-chain acylcarnitines in the mouse liver. Br J Pharmacol. 2024 Aug;181(16):2750–73.
[6] Phillips PS, et al. Statin-Associated Myopathy with Normal Creatine Kinase Levels. Ann Intern Med. 2002 Oct 1;137(7):581.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (